Cannabis Distillates
The extraction and distillation of cannabinoids has come quite a long way since its outbreak just a few years ago.
Simple extraction techniques were well-practiced by many people in the past.The state-of-the-art extraction of today's world, however, is anything but simple. 
Distillation is one of the oldest extraction processes. More recently, distillation has begun making waves across cannabis extraction industries. It is speculated that cannabis distillates are the future cannabis extract.
What exactly are these cannabis distillates? And more importantly, why is it superior to regular cannabis extract?
What Is The Definition Of Distillate?
Distillation is scientifically defined as the purification of substances through a collection of heating and then cooling methods. The distillation’s end product is called a distillate.
 Distillation yields a cleaner and purer end concentrate in cannabinoid extracts than most of the other extraction techniques.
Distillation yields a cleaner and purer end concentrate in cannabinoid extracts than most of the other extraction techniques.
In actual fact, most researchers and enthusiasts seldom refer to the final product of the distillation of cannabinoids process, as the "The Pure."
That’s because, even though the cannabinoids have been extracted via a hydrocarbon solvent, the final distillate product is “solvent-free”, meaning it has zero parts-per-million residual solvent.
The distillate products are one of the most effective and diverse types of cannabis and hemp extracts available within the market.
What are Cannabinoid Distillates?
Top class extraction practitioners use the process of distillation to extract only the specific compounds found in the original material.
It is possible to majorly distill only CBD or THC from a hemp or cannabis plant, respectively.

Cannabis distillates are not as pure as cannabinoid isolates, for they are not as refined.
Various cannabinoids, valuable molecules and minor contaminants (terpenes and flavonoids) may still be present in cannabis distillates. It can be referred to as a precursor to isolates because it could be further refined even more to isolate a specific cannabinoid.
Research has found that the cannabinoids profiles found in distillates and remaining beneficial molecules can be highly beneficial for the body.
For example, CBD Distillates usually contain THC of some amount, which could render it as illegal in some states, whereas an isolate-based CBD contains no trace amounts of THC.
Moreso, it should be known that most distillates and other extracts, produced for improved THC effectiveness are grown from the raw marijuana plant.
In contrast, the CBD distillate is extracted, mostly, from the cultivation of hemp plants.
Hemp vs. Cannabis

Cannabidiol(CBD) distillates can be obtained from Marijuana plants from time to time. However, this only increases the possibility that the distillate results in containing high levels of THC.
This can have a significant consequences due to how CBD is completely legal for consumption in all 50 states in the US, while THC is not.
CBD distillate extracted from hemp typically contains about 90 percent or more cannabidiol. The other 10 percent of the distillate is made up of other cannabinoids, and potential unwanted molecules (errors in the distillation process).
They are typically the second purest cannabidiol supplements available on the market stock. The enhanced purity offers your end customers the luxury of not consuming much to start enjoying all of its essential benefits.
Distillates are perfect in the manufacturing of capsules, vape oils, and edibles, tinctures, simply because of its purity. Distillates are also often refined even further to make the purest cannabidiol extracts on the market, isolates.
CANNABIS DISTILLATION PROCESS:
The Process
The goal of distillates is to produce a product that has a clean slate where we can infuse the desired terpenes for desired effects.
The distillation of cannabinoids, as stated above, applies to both CBD derived from hemp, as well as THC derived from Marijuana.
There is a reason enthusiasts in the cannabis community sometimes refer to cannabis distillate as "The Pure." That's precisely what it is, a product that's virtually pure cannabinoids.
So How Are DISTILLATES Made?
Cannabinoids are refined and separated by the process of distillation at the molecular level. The goal of making cannabis distillate is to isolate cannabinoids like CBD and THC from unwanted impurities.
The whole procedure is made up of several refinement processes. The main steps of the distillation of cannabinoids are explained below;
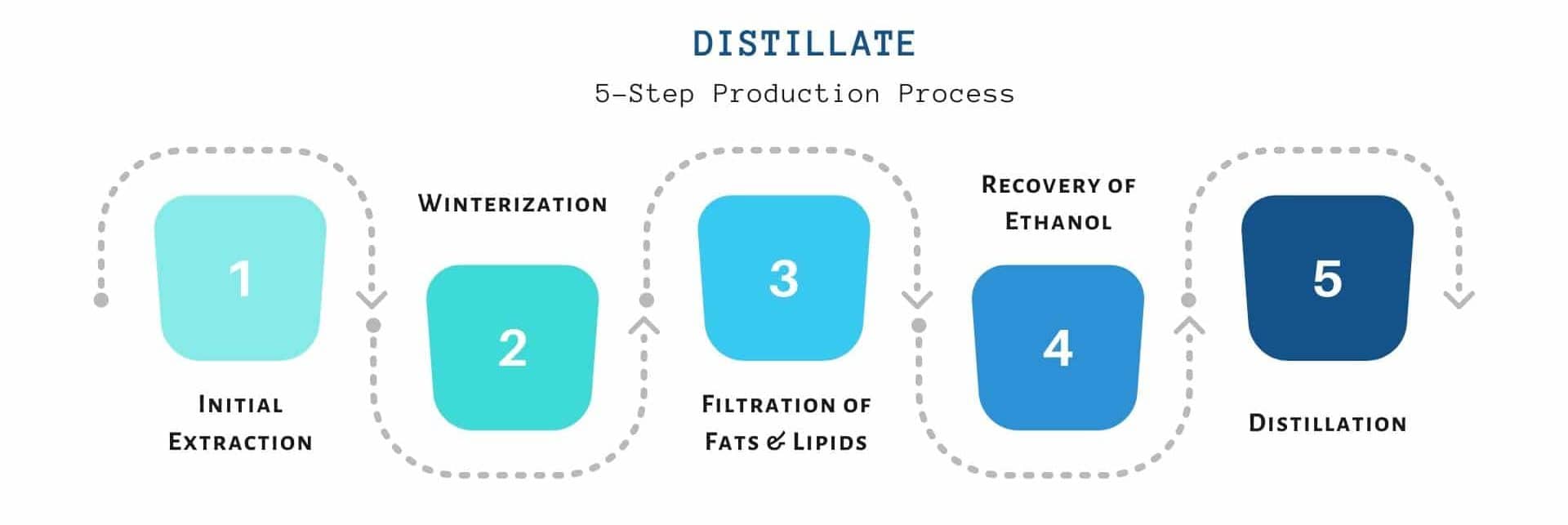
The Initial Crude Extraction of Resin from Hemp plant
The first step of distillation, the same as any cannabinoid extraction, is to extract the cannabinoids from the raw cannabis plant.
The extraction and distillation of cannabinoids are incredibly similar, as they both separate compounds.
In the initial crude oil extraction method, either solvents or physical methods (like rinsing and sifting) are used to separate the compounds.
However, there is a large difference between the two.
Distillation is separating compounds based on boiling points while extraction is separating compounds based on the solubility of the extraction solvent.
In the cannabis industry, extraction involves the collection of oils from the plant material.

In contrast, distillation describes the process of refining the extracted oil into required constituents, which can be later incorporated into marketable goods.
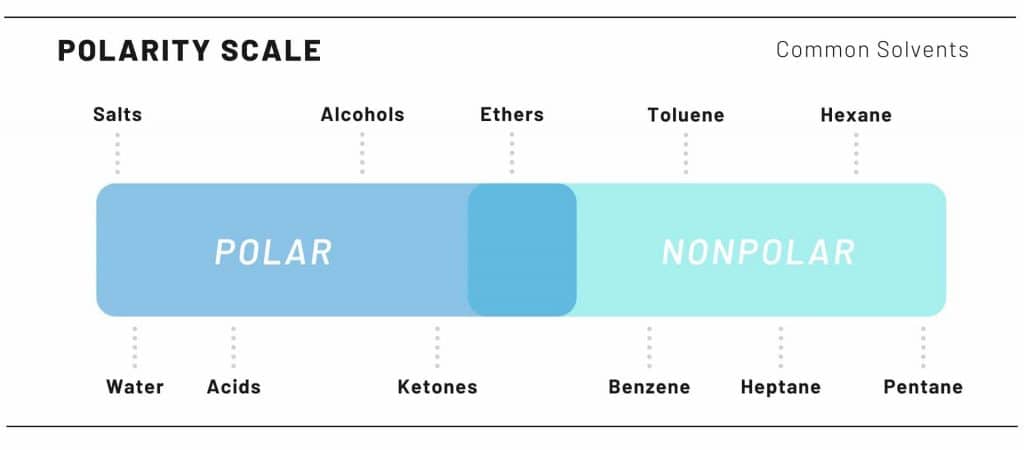
Cannabis crude oil extractions are done by polarity. Remember chemistry class? The saying “like attract likes”? Well that is precisely how solvents work to extract the cannabinoids.
Cannabinoids are nonpolar. Therefore, nonpolar solvents are used to pull the cannabinoids, leaving behind, unwanted, polar molecules. These polar molecules are things such as chlorophyll and potential water.
The polar solvents used for extractions ranges from extractor to extractor, like an artist only using a certain type of writing utilize.
Some of the more polar solvents used are Butane, Propane, & Pentane.
Alcohols & polar solvents can be used, however more unwanted product results in the final product.
The first extraction produces a product known as oleoresin, which is further refined.
The oleoresin, produced in this process, can be summed up as the concentrated cannabinoids, in addition to, all the flavoring ingredients soluble in the particular solvent used, so that it is much closer to the original...odor and flavor.
Some impurities are still present in the extracted oil - fats, lipids, and several other unwanted compounds. These compounds are removed within the next step, the winterization process.
Winterization of Oleoresin to Remove Impurities
Of course, the initial extraction process yields an extract that is full of the desired cannabinoids. But, it also contains many impurities that are unnecessary and could be harmful upon consumption.
Impurities also prevent the further steps in the distillation processes from making a high quality final product.
Winterization is conducted to remove unwanted impurities efficiently.
The winterization process is very similar to baking a chicken: In baking a chicken, the excess grease and juices (aka impurities) drip down into the pan and then thicken when cooled.
In the winterization process, the crude extract and ethanol solution is passed through a filter. Here, the fats and lipids are removed. The remaining ethanol solution is then typically recovered, done using a rotary evaporation technique.
The process starts by mixing the initial crude extract with pure ethanol.
This solution is placed in a freezing environment for roughly 24 to 48 hours at this point. During the freezing phase, most of the unwanted impurities solidify. 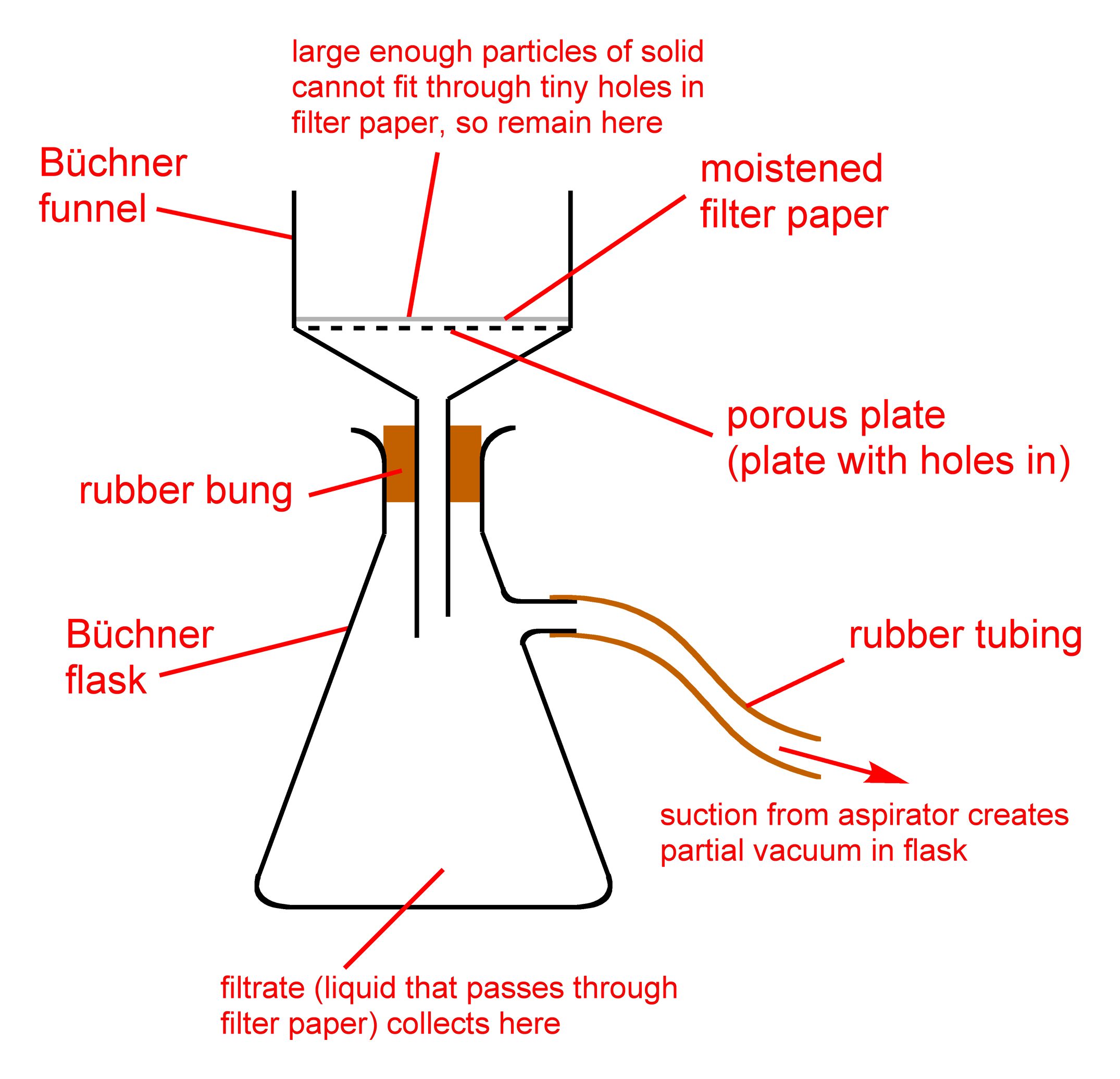
The extremely cold crude extract and ethanol solution is then passed through a filter. Fats and lipids are caught in the filter, while the cannabnoid rich ethanol passes through. This filtration process is normally performed using a simple buchner funnel.
Recovery Of Ethanol
After the fats, lipids and other minor impurities are filtered out, the ethanol is removed/recovered. Ethanol removal is typically done using a rotary evaporator.

Rotary Evaporation will remove a majority of the ethanol initially added. "One reason that rotary evaporation is favorable for this technique is that the process is efficient with minimal labor. Because the ethanol can be reclaimed once extracted in a rotary evaporator, a manufacturer significantly reduces overhead costs."
The final product is a resin containing the desired cannabinoids, other minor cannabinoids, terpenes, and antioxidants.
Decarboxylation of Cannabinoid acids
Cannabinoids found in the cannabis plant are always locked in an acidic form.
Cannabinoids generally has more benefits when changed to a more active and stable state. The is accomplished through the decarboxylation process.
Decarboxylation involves heating the solution to the right temperatures until all of the desired compounds are converted, making them activated.
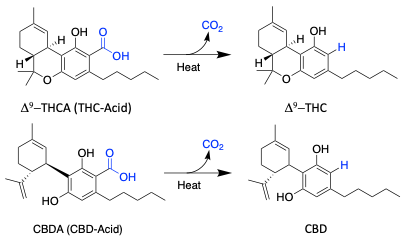
Decarboxylation of the oleoresin is done to minimize the amount of time during the actual distillation step.
However, decarboxylation can occur during the distillation process.
Either done before or after, does not seem to matter, but due to the high temperatures required during distillation, 100% of distillate is decarboxylated. Making them perfect to cook with.
Distillation
The distillation process is a separation technique that uses heat, steam, and vacuum pressure to separate compounds in a solution from each other.
Extractors can collect the compounds individually by influencing the boiling points of each compound.

In a normal cannabinoid distillation, flavonoids, terpenes, and other unwanted molecules are removed first, as they contain lower boiling points than cannabinoids.
The end product, distillate, is a golden yellow color with a viscosity that is higher than honey.
The distillation procedure can be run multiple times to get the distillates further refined.
It is important to note that there are several different forms of distillations, which are described below.
TYPE OF DISTILLATION
Simple Distillation Vs Fractional Distillation.
Simple Distillation
Distillations all have similarities in the fact that they both separate mixtures, using similar equipement. However, there are two main types of distillations which distillates are produced by.
The difference between simple and fractional distillation, at its core, is the number of times that the liquid is vaporized and condensed.
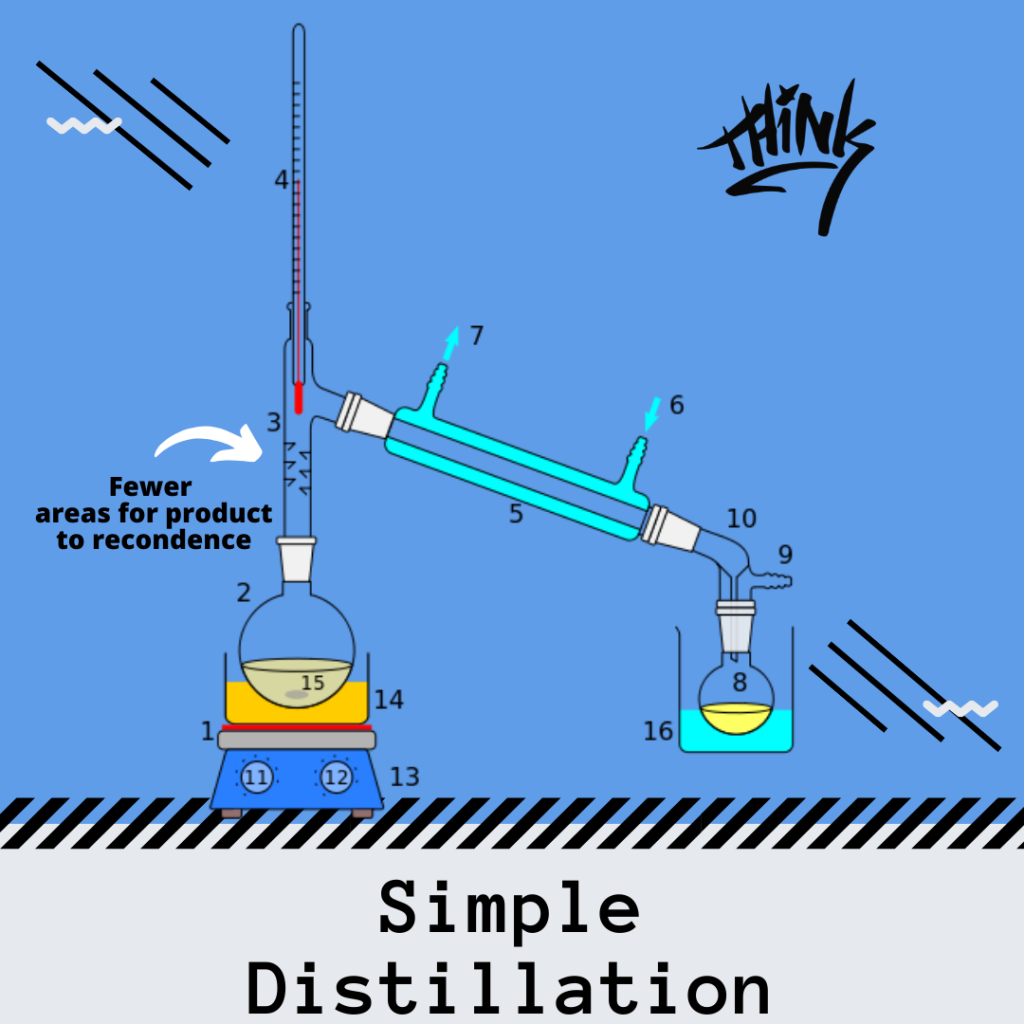
During simple distillation the liquid condenses once. Therefore a large difference in boiling points of the two liquids must be achieved to make an efficient distillation.
For example, simple distillation is often used to separate the liquid substance from a solid substance.
Fractional Distillation
During fractional distillation, two or more cycles of distillation occur.
An additional piece of equipment is used during fractional distillation, a fractionating column. This fractionating column acts as an obstruction to the gases.

As material is heated and boiled, the “not pure enough” vapors condense on these columns, falling down until it is reheated by other rising gases. This process of heating, condensing, heating, is referred to as rectification. Rectification leads to better separations leading to a purer product.
The heating and condensing process occurs until the vapor can pass through all the “obstructions” in vapor form where it is eventually collected into a flask.
Fractional distillation is commonly used when the material being distilled contains molecules with boiling points that are similar to each other.
Due to the large number of molecules within the Cannabis plant fractional distillation is the main form used to produce distillate.
Different Fractional Distillation Setups

There are multiple different fractional distillation setups. Again some cater to speed while others cater to purity.
The popular setups for distillate production currently in the industry are:
- Short Path Wiped Film Distillation
- Short Path Distillation
- Spinning Band Distillation
For this article we will specifically describe the short path distillation setup.
What is short-path distillation?
Short-path distillation is a type of technique and apparatus. It’s a simple technique where the the short-path setup involves minimal travel distance for the vapors, hence the term short-path.
The technique is used in several types of industries and has been around for over 30 years in several production fields.
This distillation is completed under vacuum, which decreases the liquid's boiling point.
Due to the decreased boiling point, it is an excellent method to be implored for unstable compounds at high temperatures or to purify small amounts of a compound.
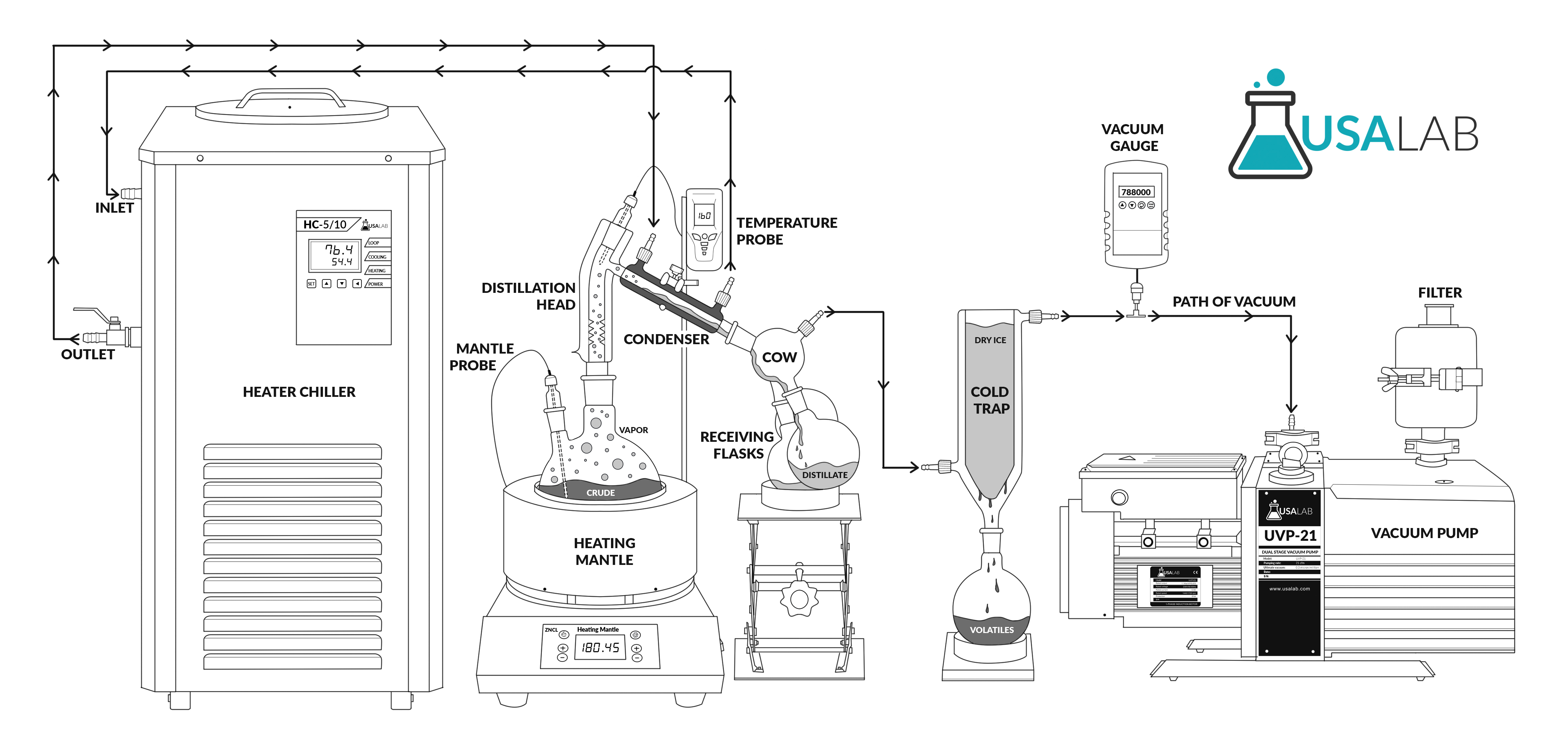
Once vacuum is applied, a flask containing the starting material is heated and boiled. As this material gradually heats up, the vapors climb the fractional column atop the flask.
As the vapors become purer and purer during the rectification stage, they eventually make their way to the condensing tube. The condensing tube is where the vapor fractions condense back into a liquid form.
The contents flow down a specific path to separate, desired flasks.
When is short-path distillation applied?
The general distillation processes in the laboratory can be elaborate and complex.
The short-path strategy utilizes glassware, which in turn, reduces potential loss of materials adhering to the inner surfaces of the apparatus.

The short-path distillation technique is employed where product loss can be expensive during production.
At an average price between $40-$70 per gram, the short path technique is a proper route to minimize costly losses.
For these reasons short path technique is a perfect fit for the distillation of cannabinoids.
All because of temperature?
The crucial part of purification by distillation is temperature.
Other physical chemistry properties contribute to the effectiveness and efficiency of the process, but the temperature is highly essential to achieving high yields of the desired products.
Defined specifically, temperature is just a measure of the average kinetic energy of the particles in an object.
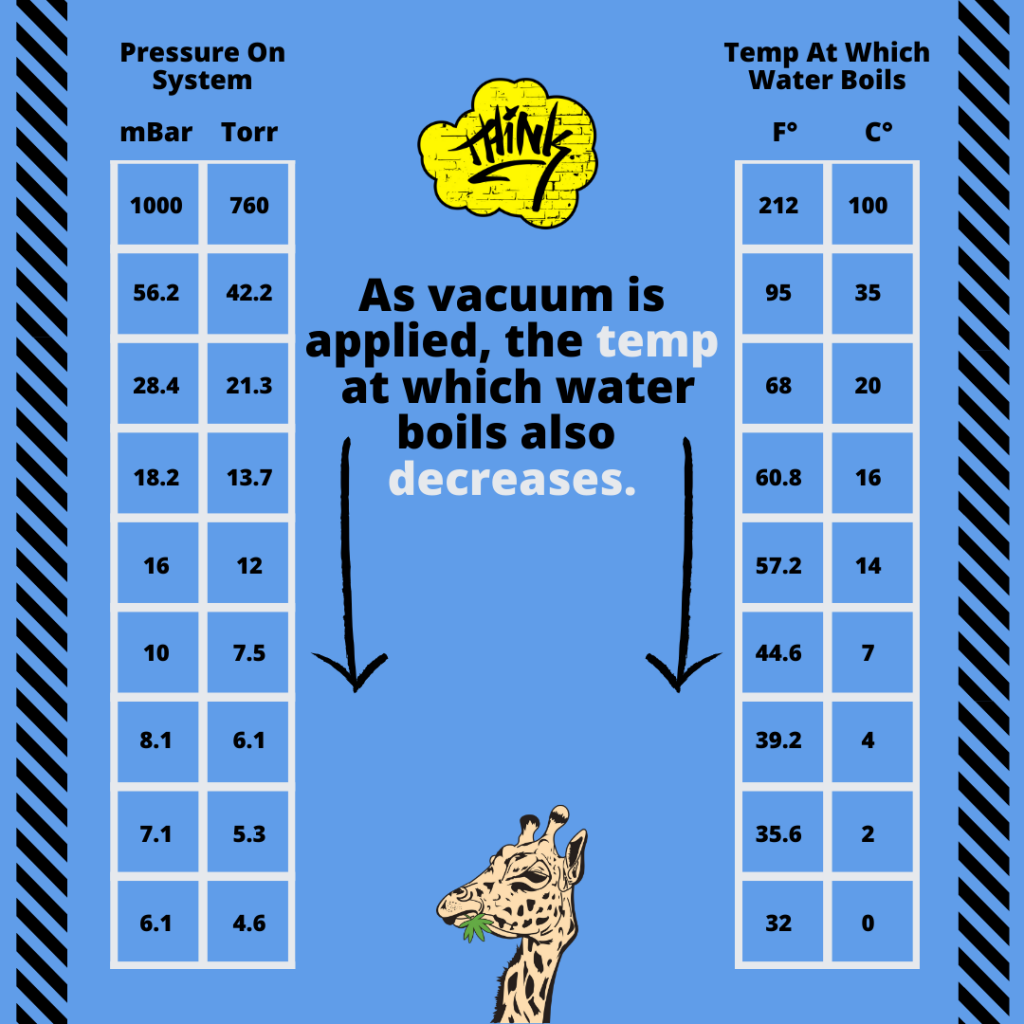
A vacuum is often used to improve the process by allowing distillation at lower temperatures, thereby reducing the potential of altering the chemical composition of distillates.
It is important to note that a vacuum doesn’t affect temperature, however it will affect the TRANSFER of heat.
This way, the molecules are not subjected to heat for a long period of time, thereby reducing the potential of altering the chemical composition of distillates.
Short- path distillation, or other forms of fractional distillation is a very important step to get the end product, without terpenoids, flavonoids, and contaminates.
Contaminants are by-products like residual solvents and pesticides. Certain pesticides do share overlapping boiling points with some cannabinoids.
A very harmful and dangerous pesticide, Myclobutanil, has a boiling point of 205°C.
CBC and THCV both have a boiling point of 220°C or over.
Therefore subjecting myclobutanil ridden product to distillation will cause pesticides to get into the final product.
The final product then has to undergo post-processing techniques such using adsorbents during flash chromatography.
HOW IS THE PRODUCTION OF DISTILLATE DIFFERENT FROM THE EXTRACTION OF OTHER EXTRACTS?
Solvent-based Vs Solventless Vs Solvent-free
The production of distillate is a bit different than the previous ways of producing cannabis concentrates such as wax, shatter, budder, and crumble. These “previous” extracts were primarily made via these two processes:
- The Solvent-based Extraction

- The Solventless Extraction
Like almost everything in the world, both extraction processes have their pros and cons. Note that neither of these methods results in pure and clean end solutions as distillation does.
Solvent-Based Extractions
A solvent-based extraction uses solutions such as alcohol or butane to pull out the wanted compounds -such as terpenes, cannabinoids, and flavonoids- from the initial cannabis plant material. Wax, shatter, and other concentrates found here are made from this method.
Solventless Extractions
A solventless extraction uses heat, pressure, or various physical rinsing methods to collect the same desired compounds. These types of extractions yield concentrates, such as bubble hash and rosin.
Solvent-Free Products
Important to not confuse solvent-free products with solventless extractions. Solvent-based extraction products can be considered solvent-free products if they undergo post extraction procedures. These post extraction procedures remove all residual solvents, used in the original extraction.

What makes THC and CBD distillate so useful?
Distillates are cannabis in one of its purest forms. On average, cannabis distillates possess around 90 percent CBD and THC from hemp and Marijuana, respectively. The rest of the 10 percent comprises other cannabinoids, and various beneficial molecules.
Cannabis distillates high concentration is one reason why it's one of the most effective cannabis products currently on the market.
Whether you're a medical patient that uses CBD or THC for its therapeutic benefits or are merely interested in enjoying recreational cannabis, distillate brings a powerful, potent punch.
You can use far lesser amounts of product to get similar results.
-
QUICK EFFECTS
The effects of distillates are felt almost immediately. In the preparation process, the THC already becomes activated (decarboxylated), and its high potency brings speedy results to patients requiring heavy-duty medication, like spinal injury, Crohn's, and cancer.
-
SOLVENT-FREE
This process creates a very pure cannabis extract and skips the need for solvents to remove any marijuana components. Extracts such as BHO (butane hash oil) and CO2 (carbon dioxide) require solvents, or alcohol-based solutions to make their end product.
Distillate is subjected to such high heat, all solvents are removed during distillation. A very high percentage of THC without any impurities is the major achievement of this method.
-
THC CONCENTRATION
This process allows between 90-99% THC in the finished product, compared to 15-25% flower THC content and 60-80% of BHO and Co2 Extractions. This method is ultra-clean, and very potent, which explains why distillation is rapidly becoming an industry-standard product.
-
DISCRETE USAGE
The pure, distilled THC produced is odorless and flavorless, which means it can be consumed discreetly. Due to the high temperatures, all terpenes and flavonoids are removed in its making.
Distillate can be used for E-cig, edibles, vape, or even topical applications. Since THC dilates the airways, it should prove beneficial to the lungs and conditions like asthma. There is a lot of research already supporting this suggestion.
CONS
Even as cannabis distillate is one of the purest forms, you would find that it isn't without its drawbacks.
One criticism of cannabis distillate is by those that ascertain that it is the start of "engineered" synthetic cannabis. Part of this reason, is due to chances of distilling impurities such as pesticides along with the cannabinoids.
By far, the most significant criticism of this type of extracted concentrate is that it contains very few terpenes (if any) that work harmoniously with cannabinoids like THC, CBD, and others to create the entourage effect.
These thoughts, however, do not seem to stop the rising popularity of distillate products.
Cannabis distillate offers much versatility to produce various beneficial consumer products that fit a wide variety of individuals.


1
9IT2sUWK
0’XOR(if(now()=sysdate(),sleep(15),0))XOR’Z
-1; waitfor delay ‘0:0:15’ —
-5) OR 735=(SELECT 735 FROM PG_SLEEP(15))–
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
https://t.me/officials_pokerdom/3706
Your article helped me a lot, is there any more related content? Thanks!
https://t.me/dragon_money_mani/35
Interesting analysis! It’s smart to look beyond pure luck in gaming. Platforms like phlwim app download apk are shifting towards data-driven play, which could be a game-changer for serious players. KYC is key for secure withdrawals, too!
FortuneTiger4 is really fun to play, the rewards are pretty good; I have never played anything like this; you should try it! fortunetiger4
Your article helped me a lot, is there any more related content? Thanks!
Heard some buzz about lebullcasino. Decided to see what all the fuss was about. It’s alright, good selection of games. Could be a good time waster with a chance to snag some serious cash. Check it out: lebullcasino
BJ88casino is alright, been messing around on there. Nothing spectacular but got some good bonuses when I signed up. Worth a look if you like promos: bj88casino
Your article helped me a lot, is there any more related content? Thanks!
Lucky999game is pretty hype! Lots of cool options and I’ve been having a blast. Definitely give lucky999game a look.
Looking for a new bookie? I’ve been testing bet10br1, and it’s got a lot going for it. Good deals and a responsive interface. Give it a shot at bet10br1.
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me? https://www.binance.info/en-ZA/register?ref=B4EPR6J0
VN123vip? Nghe cái tên là thấy sang xịn mịn rồi đó. Không biết chất lượng thế nào, nhưng mà cứ vào thử xem sao. Biết đâu lại vớ được món hời. Let’s go vn123vip!
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://www.binance.com/pt-BR/register?ref=GJY4VW8W
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
Nhóm 2: Tập trung vào Slot Game & Casino (20 đoạn) TONY01-12
Thanks for sharing. I read many of your blog posts, cool, your blog is very good. https://accounts.binance.info/da-DK/register-person?ref=V3MG69RO
I visited various blogs but the audio feature for audio songs existing at this web site is genuinely wonderful.
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
Mọi thắc mắc của bạn tại 66b đều được giải quyết ngay lập tức nhờ đội ngũ CSKH nhiệt tình, am hiểu chuyên môn. TONY01-26
I was suggested this web site by my cousin. I’m not sure whether this post is written by him as no one else know such detailed about my trouble. You’re amazing! Thanks!
Ahaa, its good conversation on the topic of this post at this place at this website, I have read all that, so now me also commenting here.
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
Great Big Beautiful Life is a book that shines. Emily Henry writes with a brilliance that illuminates the page. The epub version allows this light to shine on your screen. It is a great way to enjoy this beautiful luminosity. The writing is bright and clear. This is a book that glows. Get the digital copy and let it light up your life. https://greatbigbeautifullifeepub.site/ Emily Henry Book Great Big Beautiful Life
**boostaro**
Boostaro is a purpose-built wellness formula created for men who want to strengthen vitality, confidence, and everyday performance.
**aquasculpt**
aquasculpt is a premium metabolism-support supplement thoughtfully developed to help promote efficient fat utilization and steadier daily energy.
Your article helped me a lot, is there any more related content? Thanks!
I am sure this piece of writing has touched all the internet visitors, its really really fastidious piece of writing on building up new webpage.
Alright, folks! Just checked out 100wincom.net. Looks pretty solid, easy to navigate. Hoping to hit some big wins soon! Check it out: 100wincom
Just tried out jlbb. It seems alright, pretty standard, nothing too crazy, but still works. Here is the link to jlbb if you want to try it out.
Just signed up on jili56login and got straight into playing. Setup was easy, and site runs well. Ready to play? Jump in here: jili56login
Hey There. I discovered your weblog the usage of msn. That is an extremely well written article. I’ll be sure to bookmark it and come back to read extra of your useful info. Thanks for the post. I will definitely comeback.
I am sure this article has touched all the internet viewers, its really really pleasant paragraph on building up new weblog.
Amazing! Its in fact amazing article, I have got much clear idea regarding from this post.
I am sure this piece of writing has touched all the internet viewers, its really really nice paragraph on building up new website.
Your article helped me a lot, is there any more related content? Thanks! https://www.binance.info/zh-CN/register?ref=WFZUU6SI
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
There’s definately a lot to find out about this subject. I love all of the points you made.
I’m not sure where you’re getting your information, but good topic. I needs to spend some time learning much more or understanding more. Thanks for great information I was looking for this information for my mission.
What’s up everyone, it’s my first go to see at this web site, and article is really fruitful in support of me, keep up posting such content.
I love what you guys are usually up too. This type of clever work and exposure! Keep up the great works guys I’ve included you guys to my own blogroll.
Greetings! Very useful advice within this article! It is the little changes that will make the largest changes. Thanks a lot for sharing!
Hi! I just would like to offer you a big thumbs up for the excellent information you have got right here on this post. I will be coming back to your web site for more soon.
Your article helped me a lot, is there any more related content? Thanks!
I’ll immediately seize your rss feed as I can not to find your e-mail subscription link or e-newsletter service. Do you have any? Kindly permit me recognise so that I could subscribe. Thanks.
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
I will right away seize your rss as I can’t to find your email subscription link or e-newsletter service. Do you have any? Kindly let me realize in order that I may just subscribe. Thanks.
I will right away snatch your rss feed as I can not in finding your e-mail subscription link or newsletter service. Do you’ve any? Kindly allow me know in order that I could subscribe. Thanks.
Ahaa, its good discussion about this post at this place at this webpage, I have read all that, so now me also commenting here.
This paragraph will help the internet viewers for building up new blog or even a weblog from start to end.
Hello, I log on to your blog on a regular basis. Your writing style is awesome, keep up the good work!
I will right away take hold of your rss as I can’t in finding your email subscription link or e-newsletter service. Do you’ve any? Kindly permit me recognise in order that I could subscribe. Thanks.
best steroid to lose fat
References:
p.mobile9.com
uk casino online
References:
https://cambodia-automotive.org/2022/08/31/caif-delegates-attended-aafs-annual-council-meeting-in-bangkok-bangkok
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
mill bay casino
References:
chatawaytours.com
steroids side effects in males
References:
http://ezproxy.cityu.edu.hk/login?url=https://ecro.fr/images/pgs/?achat_hormone_de_croissance.html
References:
Web roulette
References:
https://leeway.kr/advert-eng/?bmode=view&idx=14729209
how slot machines work
References:
presse-express.dz
casino conrad
References:
https://molina.adv.br/2024/03/regularidade-fiscal-conheca-programas-de-regularizacao/
References:
Paris casino las vegas
References:
gog999.net
References:
Burswood casino
References:
https://notes.io/ecrS4
References:
Most effective bodybuilding supplement
References:
http://39.100.117.84:3000/ensfrancisca49/8136salestracker.realitytraining.com/wiki/JavaScript-is-not-available
Your article helped me a lot, is there any more related content? Thanks! https://www.binance.com/join?ref=QCGZMHR6
References:
Emerald princess casino
References:
https://www.adpost4u.com/user/profile/4308831
References:
Natural steroids in body
References:
https://www.ksg-hoesbach.de/Hoesbacher-Nachr/index.php/;focus=STRATP_com_cm4all_wdn_Flatpress_15232835&path=&frame=STRATP_com_cm4all_wdn_Flatpress_15232835?x=entry:entry251124-120035%3Bcomments:1
References:
Novice steroid cycle
References:
https://www.koeln-flink.de/Blog-Aktuelles/index.php/;focus=STRATP_com_cm4all_wdn_Flatpress_41564155&frame=STRATP_com_cm4all_wdn_Flatpress_41564155?x=entry:entry220910-145101;comments:1
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://www.binance.com/register?ref=JW3W4Y3A
References:
Steroids pills for muscle growth
References:
https://git.cyberuk.me/lamontn4928973
References:
Can steroids cause kidney failure
References:
https://megastream.pl/@billharker4877?page=about
References:
Super trenabol review
References:
https://indianmixedwrestling.com/@hansd617383121?page=about
References:
Androgen medical definition
References:
https://git.fs.cs.uni-frankfurt.de/erwin72k967090
References:
Where can i buy steroids legally
References:
https://htlm.ru/jacobstevens16
References:
Cheap steroids for bodybuilding
References:
https://git.alderautomation.ca/benjaminkitson
References:
Get bigger without steroids
References:
https://git.paulll.cc/liamcawthorne8/liam1980/wiki/Exercise-and-Testosterone%3A-Types-of-Workouts%2C-Benefits
References:
Where to get anabolic steroids
References:
http://74.48.174.77:3000/dwaynepool8183
References:
Steroids make you gain weight
References:
https://git.mana-web.com/williefryar381/willie1993/wiki/Post-Cycle-Therapy-Testosterone-Restoration%2C-Low-Testosterone%2C-HRT
What’s up everyone, it’s my first go to see at this web site, and post
is really fruitful in support of me, keep up posting
these articles.
References:
https://web.ggather.com/orchidball7/
References:
Instant Casino Live Casino
References:
https://larsen-fenger-3.mdwrite.net/instant-casino-customer-support-get-in-touch
References:
Instant Casino Kontakt
References:
https://invastu.kz/user/librairis07/
References:
Instant Casino Konto erstellen
References:
https://deal-mueller.mdwrite.net/willkommensbonus-200
References:
Instant Casino
References:
https://may22.ru/user/findiris59/
References:
Atlantic club casino
References:
https://urlscan.io/result/019d49ac-7570-7549-ab68-71cf917890e5/
References:
Casino az
References:
http://okprint.kz/user/cirruscave32/
References:
Anabolic steroids for muscle building
References:
https://alushta-shirak.ru/user/pincell14/
References:
Mobile slot games
References:
https://pad.geolab.space/s/R9eiaIU16
References:
How much do anabolic steroids cost
References:
http://king-wifi.win//index.php?title=briggsoneil3749
References:
Effect of steroids on body
References:
https://monjournal.space/item/597604
References:
What is the function of steroids
References:
https://onyxtherapy.in/hi-tech-pharmaceuticals-anavar-on-sale-at-allstarhealth-com/
References:
What is the purpose of anabolic steroids
References:
https://ghibta.org/employer/8-best-testosterone-boosters-for-men-2026-updated/
Your article helped me a lot, is there any more related content? Thanks! https://www.binance.info/ph/register?ref=IU36GZC4
Do you want to go to Montenegro? Montenegro holidays an Adriatic holiday with pristine beaches and beautiful cities. Resorts, excursions, and active recreation. An ideal destination for travel and seaside relaxation.
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://www.binance.com/sk/register?ref=WKAGBF7Y
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
изготовление рекламных флагов https://flag-zakaz-spb.ru
Your point of view caught my eye and was very interesting. Thanks. I have a question for you. https://accounts.binance.info/ru-UA/register-person?ref=JVDCDCK4
arnold schwarzenegger steroids
References:
xypid.win
Хочешь оригинальную подушку? заказать подушку дакимакура комфорт и уют для сна. Длинная форма, мягкий наполнитель и стильные принты. Отлично подходит для отдыха и расслабления.
Нужен пластический хирург? https://plasticheskaya-hirurgiya-klinika.ru современные операции и эстетические процедуры. Опытные хирурги, безопасные методики и индивидуальный подход. Консультации, диагностика и качественный результат.
Нужна мебель? производство элитной мебели эксклюзивные изделия из натурального дерева. Индивидуальный дизайн, качественные материалы и точное изготовление. Решения для дома и бизнеса.
References:
Online Casino Echtgeld 10 Euro
References:
https://kappel-melvin-2.federatedjournals.com/echtgeld-and-gratis-spiele
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me? https://accounts.binance.com/tr/register?ref=MST5ZREF
References:
Basic strategy blackjack
References:
https://mmcon.sakura.ne.jp:443/mmwiki/index.php?shirttuna96
References:
Royal vegas flash casino
References:
https://diego-maradona-ar.org/user/refundcongo4/
Today’s horoscope https://t.me/s/ulduz_fali daily forecasts and life surprises for all zodiac signs. Love, career, finances, and mood. Discover the future every day.
мебель на заказ производитель мебели из массива
References:
Rocketplay online casino neosurf
References:
https://rentry.co/gvnqpohc
References:
Masteron steroids
References:
https://peatix.com/user/29378386/view
References:
Echtgeld Glücksspiel Online
References:
https://www.investagrams.com/Profile/blanke3927388
Лучшее прямо здесь: https://reklamig.ru
References:
Hardrock casino tulsa
References:
https://telegra.ph/Echtgeld-Casino-Apps-mit-Auszahlung-04-15
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article. https://accounts.binance.com/register-person?ref=IHJUI7TF
Женский портал https://lubimoy.com.ua статьи о красоте, здоровье, отношениях и саморазвитии. Полезные советы, лайфхаки и актуальные темы для женщин. Все для вдохновения и гармонии каждый день.
Онлайн женский портал https://sweaterok.com.ua мода, уход за собой, здоровье и отношения. Актуальные статьи, советы и идеи для вдохновения и улучшения качества жизни.
Туристический портал https://swiss-watches.com.ua для путешественников: направления, маршруты, советы и лайфхаки. Подбор отелей, билетов и экскурсий, идеи для отдыха и полезные рекомендации. Планируйте поездки легко и открывайте новые страны с комфортом.
Профессиональный строительный https://newhouse.kyiv.ua журнал с полезной информацией и практическими решениями. Аналитика рынка, обзоры материалов, инструкции и советы. Всё, что нужно для качественного строительства и ремонта.
Портал о дизайне https://lbook.com.ua интерьера: идеи, тренды и практические решения для дома и квартиры. Обзоры стилей, подбор мебели и материалов, советы дизайнеров. Помогаем создать уютное, функциональное и современное пространство.
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
Строительный портал https://comart.com.ua для тех, кто ценит качество и надежность. Полезные статьи, инструкции, сравнение материалов и услуг. Найдите проверенных специалистов, получите идеи для ремонта и реализуйте проекты любой сложности с максимальной выгодой.
Строительный журнал https://ukrainianpages.com.ua с актуальными новостями, трендами и экспертными материалами. Обзоры технологий, советы по ремонту и строительству, идеи для дома и бизнеса. Узнавайте о современных решениях и применяйте лучшие практики в своих проектах.
Женский журнал https://vybir.kiev.ua статьи о моде, красоте, здоровье и отношениях. Актуальные тренды, советы экспертов и вдохновение для современной женщины каждый день.
Свежие новости https://hansaray.org.ua Украины: политика, экономика, общество и события дня. Оперативная информация, аналитика и мнения экспертов. Будьте в курсе главных новостей страны и мира в удобном формате.
Женский онлайн журнал https://whoiswho.com.ua стиль, красота и здоровье. Полезные советы, лайфхаки и актуальные темы для женщин. Все о жизни, моде и саморазвитии.
Женский журнал https://womanclub.in.ua мода, уход за собой, психология и отношения. Читайте интересные статьи, находите идеи и улучшайте качество жизни.
Портал о строительстве https://kennan.kiev.ua и ремонте: идеи, технологии, обзоры и советы экспертов. Помогаем выбрать материалы, рассчитать бюджет и найти исполнителей. Удобный сервис для планирования и реализации проектов — от квартиры до загородного дома.
Строительный портал https://solution-ltd.com.ua с актуальной информацией и практическими решениями. Узнайте о новых технологиях, сравните материалы, получите советы и найдите специалистов. Сделайте ремонт или строительство проще, быстрее и выгоднее.
Лучший сайт для женщин https://musicbit.com.ua статьи о стиле, любви, здоровье и вдохновении. Найдите идеи для жизни и развития в одном месте.
Женский сайт https://fashionadvice.kyiv.ua полезная информация о здоровье, стиле, любви и карьере. Читайте актуальные статьи и находите решения для жизни.
Онлайн сайт для женщин https://elegance.kyiv.ua статьи о красоте, отношениях, семье и саморазвитии. Советы, идеи и вдохновение для повседневной жизни.
Строительный журнал https://tozak.org.ua с полезными статьями и актуальными обзорами. Освещаем современные технологии, материалы и тренды в строительстве и ремонте. Практические советы, идеи и решения для создания комфортного и надежного пространства.
Сайт для женщин https://bestwoman.kyiv.ua статьи о красоте, здоровье, отношениях и стиле жизни. Полезные советы, тренды и идеи для вдохновения. Все, что нужно современной женщине, в одном месте.
Онлайн строительный https://reklama-region.com журнал для профессионалов и частных застройщиков. Полезные статьи, разборы материалов, новинки рынка и практические рекомендации. Все о строительстве, ремонте и дизайне в удобном формате.
Онлайн женский журнал https://zhenskiy.kyiv.ua статьи о красоте, здоровье, моде и любви. Советы, тренды и полезный контент для женщин любого возраста.
Navigating the crowded short-form video landscape requires more than surface-level feature comparisons; it demands a strategic framework grounded in data and real platform mechanics. https://npprteam.shop/en/articles/tiktok/how-does-tiktok-differ-from-reels-and-shorts-for-the-brand/ equips marketing teams with the specific insights needed to build a cohesive short-form video strategy that maximizes reach, engagement, and revenue across TikTok, Instagram Reels, and YouTube Shorts. Whether your brand is testing new platforms, consolidating fragmented efforts, or scaling proven content formats, understanding where each channel excels—and where it falls short—eliminates guesswork and anchors budget decisions in platform realities. The article provides the competitive context, audience behavior patterns, and tactical recommendations that separate campaigns that merely participate from campaigns that genuinely win in the 2026 short-form video ecosystem.
Разберитесь, почему TikTok арбитраж трафика в 2026 году становится выбором медиабайеров и трафик-арбитражников во всем мире. Платформа демонстрирует беспрецедентный рост аудитории и снижение стоимости трафика по сравнению с Facebook и Google, что делает её критически важной для профессионалов vertical marketing. Статья раскрывает ключевые преимущества TikTok: алгоритмическая эффективность доставки контента, высокий engagement?среди целевых аудиторий и относительная молодость платформы как канала монетизации. Арбитражники находят здесь уникальную возможность масштабировать объёмы при сохранении адекватного CPA, что критично в условиях насыщенности традиционных каналов. Овладев пониманием этой динамики, вы получите конкурентное преимущество на рынке и сможете переориентировать бюджеты в наиболее прибыльное направление.
Cove Forest Goods Market – Pages are well structured and respond quickly for a smooth experience.
Across various e-commerce system evaluations focused on clarity, one notable platform is Trail District Gilded Market Goods which maintains a clean layout and ensures everything feels easy to browse through today, making browsing smooth, intuitive, and well organized for all users.
In comparisons of online shopping platforms emphasizing usability and clarity, a notable example is Willow Dawn Unified Atelier which delivers pages are well organized and content is easy to understand quickly, ensuring a calm, structured, and intuitive browsing experience throughout the platform.
When comparing online shopping systems focused on usability and design flow, a standout example is Harbor Stone Shopping Hub where nice layout with clear sections and straightforward navigation flow, making browsing simple, natural, and easy for all users.
Across various e-commerce UX assessments emphasizing simplicity and flow, a notable example is Willow Pebble Vendor Studio which ensures everything feels tidy and the experience is quite user friendly, delivering a calm and structured browsing environment across all pages.
Across multiple e-commerce usability comparisons, a standout example is Frost Glade Vendor Hub Vault where feels structured and simple, making it easy to explore content, making it easier for users to locate items through a clean and intuitive interface layout.
While browsing through several options, I found this user-focused shop page and I liked how everything flowed together, making navigation feel easy and natural.
Online retail guild environments perform better when they maintain consistent visual alignment and predictable navigation paths across different pages and product groupings Raven Retail Navigation Guide improving browsing efficiency – The layout feels coherent and well spaced, helping users understand where everything is located without confusion
While reviewing different digital storefront experiences for speed and usability, I found a platform that impressed me once I loaded Seaside Creative Studio – navigation felt seamless, and the site responded instantly to every click or scroll action I performed.
During a structured review of ecommerce UI designs for usability optimization and hierarchy I explored a category page featuring a href=”[https://opalgladeboutiquehall.shop/](https://opalgladeboutiquehall.shop/)” />Opal Hall Boutique Glade Network embedded in a grid layout, – The clean layout ensures everything is easy to locate and view supporting a clear and comfortable browsing experience throughout
While assessing various marketplace vendor ecosystems, I found vendor resource center link and explored its sections while comparing data across similar platforms – I felt the platform was reasonably helpful and provided a decent browsing experience overall for initial evaluation use.
As I continued browsing charity-focused and social impact platforms, I found something placed within the text local impact hope and it is a great initiative supporting community causes and building positive local engagement overall
Somewhere along my browsing journey, I encountered a well-presented retail space and it gave me a strong sense that I would return later to explore more helpful and engaging content.
pole-haus.com – Really nice design and easy browsing experience overall today here
During a routine browsing session across several topics, I noticed something that appeared unexpectedly have a look here and although I don’t know what to expect yet, it definitely seems to have a distinct and unusual presentation
During a comparative UX analysis of online retail interfaces for structure and clarity I navigated a catalog page featuring a href=”[https://iciclegrovemerchantmart.shop/](https://iciclegrovemerchantmart.shop/)” />Grove Mart Icicle Merchant Network inside a sidebar panel, – Everything feels simple and straightforward without any distractions ensuring a clean, logical layout that supports effortless browsing and user understanding
During my exploration of online design portfolios and creative websites, I found something within the text check this website and it is a platform with clean design and a smooth browsing experience that feels intuitive
In exploring multiple online retail platforms for comparative usability analysis, I reviewed several websites and noticed gallery retail listing coral harbor a structured design approach that made it easy to locate different sections, with a generally smooth and efficient browsing experience overall.
During a long browsing session where I was reviewing several interesting platforms and ideas, I noticed something placed naturally within the content visit this cool page and it seems to carry a lively tone that feels both modern and engaging to go through
reddingroyalsfc.com – Great football club updates and match info feel engaging site
As I looked through different projects aimed at helping others, I came across something embedded within the content visit this resource and it seems to reflect a strong and purposeful idea
As I continued going through various real estate websites and listings, I encountered something within the text see more here and it presents a nice layout that gives a clear idea of what is being offered without any confusion
While performing a structured review of various experimental commerce platforms for usability testing, I analyzed layout density and content grouping when I encountered Forest Hub Trade Page which provided a surprisingly clean structure – the experience felt stable, and page elements loaded in an organized and predictable manner.
As I continued going through football club websites and sports platforms, I encountered something within the text see more here and it is a club page providing engaging match updates and football information
After going through several cluttered pages, I found this tidy store hub right in the middle, and it impressed me with its speed and a layout that was easy to follow without confusion.
While going through different personal project websites and ideas, I encountered something mid-content discover more here and it offered an interesting concept that made the experience enjoyable to explore
Арт-деко легко перегрузить деталями, что делает интерьер тяжёлым и негармоничным https://medyn.su/stili-kuhon/kuhnya-v-stile-art-deko/
Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
While exploring various learning platforms and informational sites, I noticed something within the text discover more here and it seems like a valuable source of information for many individuals
https://mhp.ooo/ru/products/tafero-25mg
While going through various pet artwork and animal design platforms, I noticed something within the content discover more here and it offers adorable pet-related prints that are highly recommended for animal lovers overall
During my exploration of niche creative retail websites for general comparison, I discovered dawn canyon boutique atelier while checking different artisan platforms and the structure appeared quite organized and easy to navigate – I found myself thinking it has a lot of potential after a brief but focused look at its layout.
Искал магазин, где можно спокойно купить телефон без риска и неприятных сюрпризов. Здесь всё прошло очень ровно: быстро приняли заказ, вежливо проконсультировали и вовремя отправили покупку. Смартфон оказался именно таким, как в описании. Надёжный вариант для тех, кто ценит нормальный сервис: smartphone купить
During a UX comparison of ecommerce systems for interface clarity and navigation flow I examined a product listing page featuring a href=”[https://ambercoastmarketplace.shop/](https://ambercoastmarketplace.shop/)” />Marketplace Coast Amber Shop Exchange within a structured grid system, – I enjoyed browsing here because load times are fast and everything appears tidy making the experience smooth and easy to manage
During my exploration of healthcare and immunization information resources, I came across something within the text view vaccination site and it is a helpful vaccine portal with clear and community focused information overall
While browsing through practical advice blogs related to improving living conditions, I included discover this page in the center – the information stood out as both useful and applicable for making small but meaningful changes at home.
During my exploration of themed entertainment and immersive websites, I came across something within the text view haunted site and it presents an interesting theme that clearly stands apart from ordinary websites on the internet
During a search for high-quality granite work examples and inspiration, I stumbled upon stonework visual hub – The examples are clearly crafted with precision, and the photos emphasize the expertise and care put into every piece.
As I was going through different fashion-focused online platforms, I encountered something within the text explore this fashion page and the design is elegant with smooth navigation, giving the whole site a very refined and enjoyable feel overall
sebastianbachlive.com – Live music updates and performances from Sebastian Bach online now
References:
Indiana live casino
References:
https://graph.org/Lucky-Green-Casino-Review–Bonus-2026-04-20
As I reviewed rock music tour websites and live event update resources, I discovered Sebastian Bach live performance news included within concert information – it provides continuous updates on appearances and shows, ensuring fans can follow the artist’s active touring and stage activity
слоты предлагают крупных выигрышей с быстрым выводом средств: casino-playfortuna
While browsing through fine arts and cultural engagement websites, I noticed something mid-content check art community and it is an art focused platform promoting creativity, exhibitions, and events overall
https://www.pravdadaily.com.ua/brendovi-podarunky-iak-vybraty-idealnyy-korporatyvnyy-prezent-dlia-kliientiv-i-partneriv/
During research into global charitable trusts and community support foundations, I came across material containing community project catalyst hub within broader discussions of social investment – it focuses on funding initiatives that drive local development, improve quality of life, and support long-term community growth through structured financial assistance
thepaleomomconsulting.com – Nutrition consulting site focused on paleo lifestyle guidance for clients
While going through different life story and inspirational narrative platforms, I encountered something mid-content visit this stories page and it is an inspiring storytelling site sharing powerful personal experiences across many lives
As I reviewed nutrition consulting and lifestyle coaching websites, I discovered a section featuring paleo wellness and nutrition hub integrated into health content – this platform provides expert guidance on adopting paleo dietary principles and supports clients in achieving sustainable wellness through personalized coaching strategies
As I explored different travel websites and hidden gem accommodations, I stumbled upon hawaii getaway hub – The charm of the place really comes through, and it has me seriously considering booking a flight to Hawaii soon.
While exploring artistic travel photography collections and storytelling websites, I came across a section featuring journey captured in images hub within creative visual content – it showcases travel experiences through photography that highlights landscapes, cultures, and personal exploration in a storytelling-driven artistic format
While browsing through unusual retail concepts and niche online store ideas, I came across odd retail concept page – The name initially feels a bit strange and unexpected, but after looking around more, the idea behind it actually starts to make a surprising amount of sense.
tribe-jewelry.com – Jewelry brand offering unique handmade designs and collections for customers
During browsing of jewelry design platforms and artisan craft stores, I came across material including handmade elegance jewelry store embedded in curated product showcases – it focuses on offering original handcrafted jewelry pieces that reflect artistic craftsmanship, cultural inspiration, and personalized design styles for fashion-conscious customers
What immediately caught my eye about this clean website – Is the logical flow of the layout, which turns what could be a messy experience into something smooth and even relaxing.
While exploring multiple sites that felt cluttered and hard to navigate, I eventually reached a thoughtfully designed marketplace and I liked how organized everything seemed, making it much easier to browse comfortably.
https://tozak.org.ua/ru/2026/01/iskusstvo-cifrovogo-otklika-kak-prevratit-poisk-raboty-v-uvlekatelnoe-puteshestvie/
While exploring different themed online platforms and local pages, I came across something embedded mid-way view this site and it has a unique feel that makes checking out its content an interesting experience overall
buy cannabis online uk
While reviewing different experimental e-commerce layouts for usability and navigation clarity during a long benchmarking session across multiple demo platforms I eventually landed on a section featuring Lemon Lark Retail Corner Hub – content was neatly organized and I could quickly locate categories without any confusion, making the overall browsing experience feel smooth and pleasant.
While testing multiple digital storefront designs for UX performance and usability flow evaluation I came across a category browsing interface containing Velvet Vendor Stone Hub integrated within a structured catalog grid, – the experience felt reliable and consistent with well arranged content that made it easy to navigate through pages and understand product details clearly.
During a casual browse of quirky internet fan projects and sports-themed celebrity pages, I discovered random celeb hub – The idea is amusingly strange, and the playful mix of volleyball and pop culture gives it a funny, non-serious vibe overall.
While testing different digital retail environments for usability behavior and interface responsiveness comparison, I came across a catalog interface containing Silk Lakefront Commerce Hub inside a structured grid layout, and – the design felt clean and distraction-free, making browsing through pages smooth and visually comfortable without unnecessary complexity.
As I browsed through real estate listing platforms and home search tools, I stumbled upon property listing page – The experience feels smooth and local, with listings that seem up to date and fairly priced for people in the market.
While analyzing multiple online shopping interfaces for responsiveness and structural clarity across simulated environments I came across a category view containing QuickCart Emporium Junction which displayed well organized elements and provided a smooth browsing experience without any noticeable performance issues during testing.
As I explored family-oriented movie recommendation sites online, I stumbled upon safe films portal – It’s nice to see a guide that prioritizes safety and clarity, without pushing any hidden agendas or unnecessary content.
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
I had been browsing aimlessly until I reached a clear and simple shop page and I found the experience very smooth, with navigation that stayed easy and free of confusion.
During analysis of multiple ecommerce boutique websites for layout consistency and UX clarity, I discovered ridge harbor boutique navigation space while comparing different systems – The design felt minimal and well structured, allowing users to move through sections smoothly while maintaining a strong sense of organization and clarity.
While exploring sports recovery blogs and football training support platforms, I discovered recovery therapy hub – The integration of emotional and physical healing within football culture feels new and interesting compared to traditional sports training resources.
During a general UX review of digital shopping platforms for responsiveness and structure evaluation, I explored category feeds and found a smooth experience when viewing Rade Collective Outlet Space – everything was well organized, and pages loaded without disrupting the browsing flow.
ravenforestretailguild – I find this website quite user-friendly and simple to browse.
While browsing rental advice blogs and urban lifestyle content, I discovered urban renter hub – The guidance feels grounded and useful, offering clear tips that are especially helpful for people new to city housing and rental processes.
While casually browsing art exhibition pages, something appeared in context, see exhibition hub, and the platform feels creative with visually engaging and well structured presentation overall
During a long session of exploring old archives and informational web pages, I noticed something placed in the middle of the content, visit this old works page, and it feels like a very interesting website where I discovered useful details while going through its sections today
In the middle of exploring fun and casual websites, I encountered something mid-content explore this site and it looks interesting overall, feeling like a relaxed and fun destination worth browsing
While reviewing several awareness platforms online, I noticed something embedded in the flow, learn more here, and the site presents structured and easy to understand educational information overall
In evaluations of digital commerce platforms focused on usability, a strong example is Dawn Willow Market Atelier where pages are well organized and content is easy to understand quickly, allowing users to navigate efficiently through clean and logically arranged sections.
During a routine search across themed entertainment websites, I noticed something embedded in content, go to site, and it delivers a spooky but enjoyable experience for visitors overall
In comparisons of modern commerce systems focused on usability and organization, a strong example is Stone Boutique Harbor Hub which maintains nice layout with clear sections and straightforward navigation flow, providing a clean and well structured browsing experience across all pages.
gay porn movies
At first my browsing felt a bit scattered and unorganized, but in the middle I discovered this structured shop page and it stood out because the content and layout felt well balanced and created a positive overall impression.
Parents who want to supplement school learning at home frequently look for organized online tools that provide inspiration and structured lessons, and they may come across child education hub listed among useful learning references – This version shows how digital platforms can support balanced educational growth through simple and engaging materials.
As I continued exploring various online portfolio and personal branding platforms, I noticed something embedded in the content learn more here and it has a clean professional design that feels simple, elegant, and well organized overall
In studies of digital storefront platforms focused on structure and clarity, a strong example is Willow Pebble Trade Studio which ensures everything feels tidy and the experience is quite user friendly, offering a smooth and logically arranged browsing experience throughout.
Many families looking for organized medical resources appreciate digital platforms that simplify complex vaccination details, and during such searches they may come across community vaccine guide featured in informational directories – The site is often described as straightforward and supportive, helping users navigate immunization choices and local health recommendations with ease.
At one point during my browsing session, I encountered something in context, visit and explore, and the design feels modern and smooth, making it very easy for visitors to navigate and enjoy the experience
Those interested in environmental appreciation frequently browse digital spaces that highlight the beauty of forests and open landscapes, and they may find nature harmony hub – The platform is often seen as a source of inspiration that promotes balance, calm thinking, and deeper connection with outdoor environments.
As I was reviewing different pet print and animal art websites, I found something embedded in the text visit dog print site and it features adorable pet-related prints that are highly recommended for animal lovers everywhere
I was casually going through various ukulele and music tutorial websites when something stood out in context, take a look here, and the overall content feels solid, recently updated, and nicely presented in a way that is easy to follow
People exploring local creative opportunities often rely on websites that bring together artists, educators, and audiences in a shared cultural environment, and they may come across art community link – This site presents exhibitions, talks, and workshops designed to support collaboration and artistic growth within the community.
While browsing personal blogs online, I came across something naturally placed within the content flow, visit lifestyle blog, and the writing feels relatable with easy to connect storytelling that keeps things simple and engaging overall
While going through seasonal event resources, I discovered find out more – The site offers an enjoyable browsing experience with clear, helpful content that feels relevant and easy to digest for all types of visitors.
While reviewing multiple travel inspiration platforms online, I stumbled upon something embedded naturally in the flow, explore this travel site, and it is a nice website where everything is organized clearly and easy to find and understand quickly
During my exploration of ecological conservation and environmental awareness resources, I came across something within the text view nature site and it is a nature focused organization encouraging environmental awareness and ongoing conservation efforts
In evaluations of online retail systems focused on simplicity and performance, a strong example is Lemon Brook Market Corner where easy to navigate and everything is clearly presented without clutter, helping users move through categories in a smooth and structured way.
mom incest porn
As I continued exploring baking and dessert recipe platforms, I found something naturally embedded in context, discover this club, and I like the platform overall because it feels reliable and easy to navigate for all users
As I continued exploring various election campaign and community leadership platforms, I noticed something embedded in the content learn more here and it is a political campaign website with clear messaging and locally engaged priorities
Voters exploring political content online often use campaign sites to gain better insight into candidate priorities and messaging congressional campaign page – The page presents structured campaign information that helps users follow policy discussions and evaluate election perspectives with ease online
While reviewing different creative arts and public exhibition websites, I found something placed in the middle take a look here and it is an art focused community platform promoting creativity, engagement, and cultural events
While casually browsing a variety of dessert baking websites, something appeared in context, see details, and I like the platform since it feels reliable and easy to navigate with clear organization
As I browsed performance focused web platforms, I encountered view hiperfree simple hub – The design is clean and minimal, with fast loading pages and smooth functionality that makes the overall experience easy and efficient.
While going through various public transit route and mobility platforms, I noticed something within the content discover more here and it provides transport information useful for commuters and travelers daily
mitchwantssununu.com – Interesting concept site, content feels direct and somewhat thought provoking today
During my search for renewable energy topics online, I noticed check this fuel project site – The content is quite engaging, and I ended up learning something new simply by exploring it for a short time without expecting much.
People who enjoy traditional style online stores often browse sites like Cove Wheat Outpost Collective where product displays are simple and well structured – The rustic design approach makes navigation intuitive and enjoyable, creating a relaxed browsing environment that feels consistent and easy to follow.
While going through various winter event listings, I came upon visit this page – The overall browsing experience feels pleasant and useful, with content that is easy to understand and structured in a way that keeps things interesting.
Across various digital marketplace studies emphasizing clarity, a strong example is Frost Glade Experience Vault where feels structured and simple, making it easy to explore content, allowing users to browse comfortably through well organized and visually balanced pages.
People who appreciate organized online marketplaces often browse platforms like Cove District Sun Goods Center where items are presented in a bright and structured layout – The design makes browsing feel smooth and enjoyable, allowing users to explore products easily without distraction or unnecessary complexity.
In the middle of researching sustainable fuel options, I came across browse this energy resource – It is an interesting platform, and I discovered new information simply by exploring it without any deep research plan.
While exploring curated listings of Hawaiian retreats and boutique inns, I discovered a highly informative accommodation page worth viewing today < coffee belt inn listing – The presentation is clear and calming, giving useful insight into the property’s atmosphere and overall guest experience very appealing
While exploring digital opinion spaces I found a minimal website that presents viewpoints in a structured but direct way using focused commentary log – the content often feels thought provoking and encourages readers to pause and reflect on political narratives and public issues discussed there
Shoppers who enjoy premium themed marketplaces often value layouts that combine luxury styling with practical navigation for better usability across devices gilded cove gold emporium – The design enhances visual appeal while maintaining a structured browsing flow that feels smooth, balanced, and easy to follow.
People who prefer efficient online shopping systems often engage with sites like Harbor Kettle Commerce Core Hub where items are displayed in a clean format – The design ensures browsing feels organized, practical, and easy to follow across all sections.
Digital archives dedicated to European festivals often include detailed records of music performances costumes and community participation over many years festival_history_portal which scholars use to trace how cultural events evolve maintain traditions and engage audiences through changing social contexts over decades
Across various e-commerce experience evaluations focused on usability, a notable example is Gilded Brook Network District where nice visual balance and navigation works without any confusion, helping users interact with a clean and efficient browsing environment across all pages.
As part of my research into festive online experiences, I came across explore this festival – The information is presented in a fresh and engaging way, making it enjoyable for users to browse while still feeling informative and practical.
As I looked through humorous online platforms, I noticed view comedy entertainment site – The vibe is playful and engaging, and the content feels light, making the browsing experience smooth and enjoyable overall.
Users who prefer easy to navigate ecommerce platforms often explore sites such as Cove Goods Sun District Shop Hub where products are arranged in an organized and bright layout – The browsing experience feels smooth and enjoyable, allowing users to quickly move through categories without confusion.
Users who appreciate curated artisan stores often explore platforms like Artisan Wave Market Trail where product presentation focuses on clarity and aesthetics – The design highlights each item in an organized layout, making browsing feel intuitive, visually appealing, and easy to navigate across all categories.
Users exploring modern ecommerce vault-style platforms often appreciate how structured layouts improve clarity and product discovery while maintaining a clean visual hierarchy across categories and sections Glass Harbor Vault Hub – The clean vault concept delivers a structured browsing experience where navigation feels intuitive, organized, and visually easy to follow throughout the entire platform.
modelscanvas.com – Creative portfolio vibe, visuals and layout feel clean and professional design
While browsing unique digital retail ideas, I discovered a conceptual supermarket platform that focuses heavily on simplicity and usability hope concept food hub – The layout is clean and straightforward, providing an easy browsing experience without unnecessary complexity or cluttered design elements
As I browsed various self-help and resilience websites, I encountered view this life journey site – The content feels meaningful and well considered, with a relatable tone that makes the overall reading experience feel genuine and reflective.
In comparisons of modern shopping platforms focused on user experience, a strong example is Glade Trade Night House which maintains everything feels straightforward and browsing is comfortable and stable, ensuring a calm and easy navigation flow throughout the entire site.
While reviewing several foundation pages, I found check this out – The information is presented in a straightforward format, making it easy for users to understand without needing additional explanation.
Political journalism students examining election cycles often reference candidate webpages as primary sources for understanding messaging tone and issue prioritization strategies election resource hub – The campaign platform outlines key policy areas and voter engagement updates in a manner intended to remain transparent and frequently refreshed
Users who prefer handcrafted ecommerce environments often explore sites such as Teal Vendor Cove Creative Atelier Hub where products are presented in a stylish and artistic layout – The interface creates a visually engaging browsing experience that feels structured, modern, and thoughtfully curated for easy exploration.
While casually browsing a variety of informational links, something appeared that stood out slightly, see details, and it gives a fair amount of information that is simple to read and follow without complications
Users who appreciate structured creative hubs often browse sites such as Vendor Apricot Works Meadow Hub Portal where content is organized neatly – The design ensures simple navigation, clear presentation, and accessible browsing experience.
While searching for creative showcase examples I came across a modern portfolio site that highlights visuals with clarity including artistic portfolio gallery – the interface feels professional and well structured offering a clean and visually appealing browsing experience
Individuals who prefer clean digital retail environments often gravitate toward platforms that emphasize structure, especially when they discover sites like Berry Cove Digital Store where digital presentation is carefully arranged to support smooth navigation and easy understanding of product offerings – The store maintains a polished digital layout that supports clarity and efficient browsing across all pages
As I browsed various online offers and resources, I encountered view this bonus resource page – I found it today and it appears useful overall, with a nicely presented layout that feels simple and easy to navigate.
While researching luxury beverage brands online, I came across a beautifully designed winery website that emphasizes both product detail and brand identity iniskillin wine detail portal – The wine information is detailed and appealing, creating a professional and polished overall browsing experience
While scanning through various opinion and commentary pages, I came across something in the content flow, click to view, and it works fine overall with a clean design and user-friendly layout that is simple to use
Across various UX evaluations of digital marketplaces, a strong example is Sage Harbor Experience Vault where clean design and content is arranged in a logical order, allowing users to browse comfortably through well balanced and structured pages.
While analyzing different environmental awareness websites, I noticed use this resource – The layout is clean and structured, making it easy for users to appreciate the effort and care put into presenting the information clearly.
Users who appreciate streamlined ecommerce environments often browse platforms such as Teal Harbor Commerce Flow Hub where products are grouped in a clean and structured format – The design makes browsing feel smooth, efficient, and easy to navigate, with clear categories guiding user exploration.
While exploring social progress organizations, I encountered explore this empowerment network site – The initiative appears meaningful, and the information is clearly structured, making it easy to understand and navigate.
Shoppers who value structured ecommerce environments often appreciate clean collective systems that make browsing intuitive and visually balanced across all pages Gladeridge Collective Goods Hub – The layout is modern and minimal, providing a smooth browsing experience where products are easy to locate and visually well arranged.
Community health advocates and social workers frequently refer to local support initiatives when mapping assistance resources for vulnerable populations compassion_hub that prioritize dignity centered care and accessible outreach programs across different neighborhoods and service areas – It emphasizes ongoing compassionate services and structured community engagement designed to support people facing hardship in practical ways
While browsing classic theme websites I discovered a nostalgic platform that focuses on visual engagement featuring retro themed showcase page – the overall presentation feels cohesive and makes browsing feel enjoyable and visually appealing
I didn’t expect much while browsing randomly, but then something appeared that caught my attention, check more info, and my initial impression is positive because the design feels modern, clean, and very well put together
As I continued exploring lifestyle and empowerment websites, I found something naturally embedded in context, discover this site, and it turned out to be really helpful where I gained useful insights and enjoyed browsing today
While exploring experimental online design concepts, I found a platform that stands out through its unconventional and creative structural layout digital abstract structure hub – The content feels thoughtfully experimental, with a layout that encourages exploration and highlights creative presentation techniques
As I continued going through various nonprofit housing assistance websites, I encountered something within the text see more here and it is a support organization focused on housing aid and essential services
Shoppers who value handcrafted digital stores often prefer platforms such as Brook Opal Artisan Market where design and product presentation emphasize originality and craftsmanship – The overall experience feels curated and genuine, helping users easily explore artisan goods while maintaining a calm and structured browsing flow.
Across multiple online shopping comparisons emphasizing usability, a standout platform is Amber Summit Experience Marketplace where smooth experience overall, pages feel fast and easy to use, making browsing feel natural, calm, and highly intuitive across all sections.
As I explored various online squad platforms, I encountered view squad digital hub – The site looks visually impressive, and the content remains interesting, making the overall experience smooth and enjoyable.
While going through various musician pages, I discovered find out more – The layout feels straightforward and easy to use, helping visitors quickly understand and navigate the site with ease.
Users who prefer minimal ecommerce systems often explore sites such as Trail Commerce Harbor Market Hub where products are grouped in a clean and accessible format – The design ensures browsing feels smooth, user friendly, and well organized, helping users quickly locate what they need without unnecessary complexity.
Shoppers who value modern digital storefronts often prefer emporium designs that enhance usability through structured visuals and clear layout hierarchy Harbor Glass Emporium View – The presentation is sleek and consistent, making browsing feel smooth, visually appealing, and easy to understand at every step.
nomeansnoshow.com – Strong identity here, site feels bold and creatively expressive throughout pages
During my exploration of structured web pages, I noticed check blunty content hub – The layout is clean and well organized, making navigation smooth and easy to follow while going through the different sections.
While scanning through lifestyle and hospitality pages, something caught my attention in context, click hospitality site, and the platform feels lively and engaging with a user friendly browsing experience overall
While browsing digital resume and portfolio pages online, I discovered a clean profile website that feels highly professional and easy to use professional identity showcase hub – The layout is smooth, with clearly structured content that is simple to understand and well organized throughout
When analyzing e-commerce platforms built for usability and structure, one standout example is Icicle Merchant Lakefront Mart where simple layout and information is easy to find at a glance, helping users access content without confusion or unnecessary clutter.
While researching modern marketplaces with soft and elegant aesthetics, I explored browse this willow velvet hub – The layout is gentle and refined, and users can navigate easily while enjoying a smooth and peaceful browsing flow.
Many online shoppers exploring curated handmade marketplaces often notice how presentation influences trust when browsing stores such as Artisan Caramel Trail Shop where warm visual styling and organized categories help guide the experience – The overall aesthetic emphasizes a cozy handcrafted feel while maintaining structured product layouts that make browsing simple and inviting
People who appreciate beach themed digital marketplaces often browse platforms like Coastal Wave Harbor Outpost Hub where items are displayed in a soft and minimal design – The layout ensures browsing feels calm, pleasant, and visually balanced, making product discovery easy and enjoyable.
While searching through celebration resources online, I encountered access this page – The content is presented consistently well, with an engaging tone that makes the overall browsing experience smooth and enjoyable.
Очень удобный сервис для покупки телефона онлайн. На сайте легко найти нужную модель, а после заказа сотрудники быстро выходят на связь. Доставка аккуратная и без задержек, сам смартфон полностью соответствует ожиданиям. Хороший магазин для спокойной покупки: stor 77 интернет магазин
Shoppers exploring modern ecommerce environments often enjoy emporium layouts that balance strong visual identity with functional navigation across all sections Stone Glass Emporium View – The design is consistent and minimal, ensuring browsing feels clear, structured, and visually easy to process at every stage.
As I explored various online commerce hubs, I checked see meadow linen commerce hub page – The experience is simple and pleasant, and browsing feels smooth and enjoyable with clear organization across the site.
While exploring creative portfolios and expressive platforms I discovered a site that emphasizes bold design choices including art driven web hub – the site feels confident and visually rich providing an engaging browsing journey from start to finish
During my exploration of niche online ideas, I stumbled upon check this creative clone page – It stands out as something different and is definitely worth checking out for its originality and design concept.
Across multiple usability tests of retail systems, a notable example is Orchard Global Upland Hub which maintains well structured pages and browsing feels natural and efficient, helping users find content quickly with minimal effort.
While browsing cross cultural digital platforms, I encountered a site that blends artistic and global influences into a visually engaging layout world culture fusion site – The website feels interesting and diverse, combining themes in a way that creates a rich and engaging user experience overall
Users who appreciate artisan marketplace structure often browse platforms such as Wind Cove Artisan Supply Market Hub where products are arranged clearly for easy browsing – The design ensures browsing feels intuitive, organized, and visually pleasant throughout the store experience.
While comparing retail atelier systems for usability and holiday appeal, I discovered browse mint orchard market hub – This is definitely somewhere I’ll come back to for the holiday season because the experience feels easy and enjoyable.
During my browsing of professional development and business help platforms, I noticed several informative pages and found professional help desk page – The information is clearly presented and practical, helping users quickly understand solutions without unnecessary complexity or confusion.
Artisan inspired ecommerce platforms often focus on handcrafted presentation styles and visually rich browsing environments Galleria Cove Artisan View – We design each section to emphasize clarity and storytelling using structured layouts soft visual transitions and consistent design language that helps users navigate comfortably while appreciating detailed product presentation and maintaining engagement throughout their browsing journey experience overall platform today
As I explored various trust-oriented platforms, I encountered learn more here – The information is presented in a clear and structured way, making it feel reliable and valuable for users seeking straightforward and meaningful details.
oakmeadowcommercehub – Commerce hub feels organized, categories are clear and easy browsing
nutschassociates.com – Professional services look solid, information is clear and easy to follow
Online craft enthusiasts often browse diverse digital stores seeking inspiration and quality handmade items while occasionally discovering platforms such as violet harbor artisan gateway offering a wide selection of creative goods and unique browsing categories for relaxed shopping sessions – The marketplace emphasizes smooth navigation and a pleasant user experience focused on handcrafted variety.
At one point during my browsing routine, I noticed something that appeared naturally within the flow of content, open this page, and it offers interesting information that makes me think I will definitely visit again sometime soon
“Сервис [url=https://selector-casino.cm/]selector casino[/url] порадовало дизайном.
Здесь понятно играть.
Выбор игр действительно полный.
Присутствуют бонусы — всё усиливает игровой опыт.”
While searching through work support systems and online guidance tools, I found useful materials and encountered work support network hub – The platform provides clear and practical insights, making it easy for users to understand and use the information efficiently.
uplandcovevendorcorner – Vendor corner feels helpful easy browsing and clean layout overall
Across multiple online retail usability analyses, a notable example is Lakefront Frost Global Vault which ensures clean interface and everything is easy to navigate without effort, delivering a seamless and well organized browsing journey across all pages.
People who enjoy organized digital outlet stores often browse sites like Stone Harbor Outlet Commerce Hub where products are grouped logically – The design ensures fast navigation and easy access to items, creating a straightforward and clutter free browsing experience for all users.
While researching creative fusion websites, I encountered a platform that integrates cultural elements from different regions into one engaging design brooklyn jeddah theme site – The content feels diverse and interesting, with a strong blend of styles that makes the experience more dynamic and engaging overall
People who prefer peaceful aesthetic galleries often explore sites like Galleria Dawnstone Soft View where content is displayed gently – The design makes browsing feel smooth, calming, and visually easy on the eyes across categories.
While reviewing various health lifestyle resources, I encountered discover this clean eating site – The presentation feels structured and easy to navigate, making the overall experience smooth and enjoyable for users.
While browsing modern enterprise websites I discovered a platform that focuses on structured presentation and ease of use using enterprise design showcase – the layout feels clean and supports smooth interaction across different sections of the site
Shoppers exploring modern trading collectives often appreciate platforms like Pine Harbor Market Collective where the structure supports ongoing seller collaboration and product variety – The design feels lively and interconnected, making the shopping experience feel more like a community marketplace than a traditional store.
While searching for nature retreat destinations, I encountered explore camping travel hub – It seems like a great place, and the information is clear, engaging, and helpful for planning outdoor stays.
развивающие занятия для детей
While scanning through personal branding websites, something caught my attention in context, click profile site, and the platform offers clear presentation of work with a professional feel overall
When comparing online retail systems focused on usability and structure, a standout example is Frost Forest Shopping Vault where the design feels balanced and content is clearly organized, making navigation simple, natural, and easy for all visitors.
Users who enjoy cool themed online stores often engage with sites such as Icicle Market Winter Isle Hub where products are arranged in a crisp and refreshing design – The layout ensures a smooth browsing experience that feels clean, structured, and visually calming across all product categories.
While searching for unique sports themed entertainment pages online I found a small but engaging volleyball focused site volley entertainment micro site – It blends humor and fandom style writing in a simple layout that keeps the experience smooth and enjoyable for casual visitors
champselyseesclinic – Clean design and helpful content, feels professional and easy to explore.
Community members and civic watchdog groups often reference campaign websites to track candidate goals and evaluate how transparently information is shared with the public voter_information_page – The platform communicates campaign priorities and engagement efforts in a structured way aimed at helping voters stay informed and connected with ongoing updates
During my casual exploration of creative media platforms, I noticed check this image page – I found it by chance, yet it turned out to be more useful than expected, presenting content that feels relevant and worth exploring further.
pair-dating.com – Dating concept looks simple, interface feels straightforward and user friendly experience
Users exploring friendly ecommerce layouts often appreciate stores such as Cove Soft Market Hub which combines gentle visual styling with practical organization – The interface allows users to browse comfortably while maintaining a strong sense of structure and clarity across all categories
During my search for holiday gala events, I noticed check this festive christmas page – The atmosphere feels very festive, and the theme and presentation style are both attractive and well structured for easy browsing.
People who value practical online stores often explore sites such as Stone Glade Outpost Utility Market where product listings are presented in a minimal layout – The interface focuses on usability and structure, making browsing feel smooth, clear, and highly functional throughout the shopping experience.
In the middle of looking through creative travel photography sites, I found explore this travel gallery – The overall experience is excellent, with quick page loads and a layout that feels easy to follow and naturally structured.
While browsing online housing platforms I came across a site dedicated to Kaufman County real estate that keeps things simple and readable local housing search center – It provides an easy navigation flow and structured listings that help users focus on finding relevant property options quickly
While exploring online platforms that emphasize connection and simplicity I noticed a website featuring dating interaction hub – the overall layout appears structured and the interface provides a smooth and easy to follow user experience from start to finish
During my exploration of parenting communities online, I stumbled upon check moms family hub – The blog feels approachable and friendly, and the content is genuinely helpful for readers navigating daily parenting life.
Users who prefer calm and minimal shopping experiences often engage positively with Ginger Vault Direct Shop which focuses on straightforward navigation and structured product listings – The interface feels secure and thoughtfully designed to support easy browsing decisions.
Users who prefer simple rustic ecommerce systems often explore sites such as Timber Trail Rustic Outpost Valley Hub where products are arranged in a warm wood themed format – The interface creates a smooth browsing experience that feels natural, easy to navigate, and visually well structured throughout the store.
Your point of view caught my eye and was very interesting. Thanks. I have a question for you. https://accounts.binance.com/register-person?ref=IHJUI7TF
piercethearrow.com – Bold branding here, content feels energetic and visually striking creative site
As I explored music-related entertainment pages, I came across view chris young entertainment site – The platform feels solid and well organized, and the information flow makes everything easy to follow while browsing.
During my exploration of online jewelry stores, I noticed check artisan jewelry page – The design and content feel nicely aligned, creating a visually pleasing and easy-to-navigate experience for users.
As I moved through different informational resources and guides, I found something that appeared naturally between everything else, explore further, and it seems like a practical site offering clear content and straightforward navigation overall
While browsing child friendly entertainment platforms I came across a site that highlights carefully selected films appropriate for families and younger viewers kids safe cinema listings – The layout feels clean and easy to navigate, with a strong focus on appropriate and well curated content
People who enjoy refined digital collectives often engage with platforms like Stone Golden Boutique Collective Hub where product presentation is carefully curated – The layout highlights elegance and structure, ensuring a smooth browsing experience that feels premium, consistent, and visually harmonious throughout the store.
While exploring modern e commerce interfaces with minimal aesthetics I discovered a platform featuring amber retail space – the calm design style makes browsing comfortable and the layout feels clean and well organized
During my browsing of online management utilities, I found check claimstrk tracking hub – The tool feels very simple and useful, making it a practical option for users looking for easy tracking solutions.
While exploring artistic web platforms I found a site centered around high energy visuals page – the interface feels lively and the design elements combine to create a strong and visually striking presentation style
In the process of reviewing multiple structured websites, I found explore this rtc site – The organization of content stands out, helping users easily locate important information without feeling overwhelmed or lost in disordered sections.
While browsing confectionery themed design platforms I discovered a website that presents sweet branding concepts in a structured and visually engaging way that makes it easy to navigate and enjoyable to view for design enthusiasts dessert branding concept page – The site feels visually appealing and neatly organized, creating a smooth and consistent user experience
While reviewing a mix of travel inspiration sites, I came across something that seemed worth noting, open and see, and it feels like a lovely hotel that stands out in a very appealing way
During my search for opinion-driven and debate-focused websites, I noticed check this bold opinions site – The material is quite attention-grabbing, and the content stands out immediately with a strong and assertive presentation approach.
People who enjoy artisan styled marketplaces often engage with sites like Trail Harbor Artisan Workshop House where products are presented in a warm and welcoming environment – The design emphasizes comfort and visual harmony, making browsing feel smooth, relaxed, and visually consistent throughout the site.
Users exploring creative online stores often appreciate platforms that simplify browsing and highlight unique items and during exploration they may find violet harbor artisan network featuring organized craft selections designed for easy discovery and enjoyable interaction – The platform focuses on delivering a smooth handcrafted shopping experience.
As part of studying modern commerce hub layouts, I explored check this meadow commerce hub – The experience feels clean and easy, and navigation is smooth, making browsing enjoyable and stress free today.
preventcovid19trial-uk.com – Informational tone here, content feels research focused and medically structured layout
People who enjoy aesthetically soft ecommerce galleries often engage with platforms like Galleria Dawnstone View where items are displayed in a gentle and minimal layout – The design creates a calm browsing atmosphere that feels visually light, balanced, and easy to navigate without distraction.
Users who value simplicity in online shopping often prefer platforms that reduce visual overload and highlight essential product details, such as Meadow Essentials Storefront where the interface focuses on clarity and structure, allowing visitors to browse comfortably without unnecessary distractions or complexity.
In comparisons of online retail systems focused on UX efficiency, a standout example is Harbor Trade Violet House where clean structure overall, makes browsing feel smooth and simple, supporting fast access to information through a well-organized interface design.
While comparing soft-themed online marketplace platforms, I discovered explore velvet willow space – The interface is elegant and smooth, and browsing feels calm, intuitive, and consistently easy across all pages.
Many users browsing curated marketplaces report that structured design enhances usability, especially when they reach Meadow Vendor Showcase Access Point and they often highlight that the system reduces confusion during navigation – the experience is described as streamlined with improved accessibility to relevant product listings throughout
While reviewing digital retail atelier marketplaces, I came across visit mint orchard artisan atelier – I will definitely be coming back here for the holiday season due to its clean design and easy navigation flow.
As I explored different yoga-related resources online, I encountered view yoga exploration page – The concept appears quite interesting and offers a perspective that could be worth studying further over time.
As I browsed fine jewelry marketplaces, I noticed view elegant diamond gallery – The site feels refined and elegant, and the presentation is polished, making it visually pleasing and well structured.
While searching sports recovery education platforms I came across a football therapy website that provides practical guidance on rehabilitation and performance care in a clear and supportive format designed for athletes and fitness enthusiasts sports therapy training hub – The site feels structured and useful, offering accessible recovery information
Users who prefer structured ecommerce outlet layouts often engage with sites such as Harbor Pine Smart Outlet Hub where items are arranged in logical sections – The design makes browsing straightforward and efficient, helping users quickly find products while maintaining a well categorized and easy to navigate interface.
Users who prefer practical ecommerce structures often engage with sites such as Kettle Harbor Online Trade Hub where products are displayed in a structured format – The interface makes browsing feel simple, smooth, and easy to explore across all sections.
While comparing commerce hub systems for usability, I discovered browse vale harbor digital commerce hub – The platform is clean and well structured, and navigation feels simple and easy for users.
During casual research on vendor directories, I found that Violet vendor hall catalog appears within a clean digital environment – it emphasizes organized product grouping, making it easier for users to scan through offerings and identify items of interest efficiently.
As I browsed through tourism discovery platforms, I noticed view this travel info ireland – The site feels engaging and user friendly, allowing easy browsing of travel details and destination highlights.
People who enjoy efficient shopping environments often prefer vault style designs that emphasize clarity structured layouts and smooth category transitions Hazel Harbor Vault Network – The interface is designed to reduce complexity while maintaining strong visual order allowing users to browse comfortably and locate products quickly within a cohesive system that prioritizes usability and simplicity throughout the entire ecommerce experience today platform.
While browsing medical research websites to understand how they present data I discovered a platform showcasing study participation portal – the content feels structured and the overall design reflects a clear and research focused approach to information delivery
velvetcoveartisanoutlet – Artisan outlet design clean, products are nicely arranged and easy to explore
uplandcovevendorcorner – Vendor corner feels helpful easy browsing and clean layout overall
While browsing various online band pages, I found explore this music site – The style is quite appealing, and I like the presentation, which feels simple, clean, and visually engaging throughout the browsing experience.
Shoppers who enjoy minimal and bright ecommerce design often browse platforms such as Harbor Sunlight Market Hub – The interface is structured for clarity and usability, allowing users to move through categories easily while enjoying a clean and welcoming shopping environment.
nightorchardretailmart.shop – Bought a gift last week, packaging felt really premium honestly.
While searching urban culture resources I came across a Seattle lifestyle platform that presents modern city living content in a visually rich and energetic way making it engaging for readers interested in urban environments urban seattle lifestyle guide – The content feels dynamic and modern, with strong visual storytelling
Users who enjoy simple online marketplaces often engage with sites such as Upland Harbor Commerce Easy Flow Hub where products are organized for quick discovery – The design ensures navigation feels intuitive, clean, and efficient across all product sections of the store.
During my review of fashion retail platforms, I encountered explore apparel style collection – The presentation feels very stylish, and the products along with content are displayed really well, making the browsing experience smooth and attractive.
As I reviewed structured boutique hall websites, I noticed check velvet brook boutique portal – I found helpful information, and everything appears organized, making it easy to follow across all sections of the site.
In exploring online handmade hubs I found a platform that blends creativity with structured and accessible design system where Walnut Cove artisan marketplace portal offering diverse artisan creations with strong visual organization layout clarity – It showcases handmade artistry while building a connected community of independent creators globally online network
People who enjoy organized online shopping environments often visit sites such as Cove Vale Goods Market District where items are arranged in a minimal structured format – The layout makes it easy to browse products, compare options, and navigate smoothly across categories.
While examining several educational trading resources for structure and readability, I found during analysis flow Trade Insight Harbor Network listed within relevant materials – revised note: information is organized cleanly, making it simple for readers to follow explanations step by step without confusion.
While exploring online retail platforms I found a shop that emphasizes clean product layouts and straightforward browsing structure making it convenient for users looking for a simple shopping experience simple ecommerce storefront – The design feels minimal and practical
During research into modern and simple eCommerce vendor halls, I explored browse this mint vendor site – The interface is uncluttered and easy to use, and browsing feels smooth and efficient.
While studying marketplace website structures, I found Birch Harbor vendor exploration page integrated into the central layout – The vendor hall offers multiple listings and provides a smooth user experience, making it easier for visitors to browse categories and understand product organization clearly.
During a detailed review session, I encountered see more info – The material is presented in a straightforward manner, making it easy to digest and leaving a strong impression of reliability and thoughtful organization.
Shoppers who appreciate stylish ecommerce experiences often browse platforms such as Trail Harbor Vendor Visual Studio where product showcasing is designed to highlight creativity and modern aesthetics – The browsing flow feels smooth and curated, offering a visually engaging environment for discovering vendor products.
As I reviewed examples of online commerce hub platforms, I checked see this velvet grove shop page – The website offers good options, and I enjoy checking listings regularly because they are clear, organized, and easy to browse through.
Online shoppers increasingly seek platforms that enhance usability and reduce browsing complexity, especially when dealing with large catalogs structured within systems such as Coastal Deals Hub which offers a streamlined interface designed to help users navigate categories efficiently, ensuring quick access to relevant products while maintaining consistency across the browsing experience.
People who appreciate modern vault themed stores often browse platforms like Ridge Vault Ivory Market Space where items are arranged in a clean and structured format – The design ensures content feels curated and visually balanced, making browsing smooth, intuitive, and aesthetically pleasing throughout the entire shopping journey.
While exploring online community resources I came across a Lochwinnoch site that shares local information in an inviting and easy to follow style helping users learn more about the area and its community services lochwinnoch local info hub – The presentation feels warm and approachable, focused on helpful details
People who appreciate curated online marketplaces often engage with sites like Harbor Teal Collective Market where items are displayed in an organized minimal layout – The design ensures browsing feels modern, intentional, and visually refined with well structured content flow.
uplandharborcraftmarketplace.shop – Navigation could improve but products are unique and cool.
As I evaluated several online marketplaces for outdoor and hiking products, I paid attention to navigation structure and usability, and in the middle of that review I came across Harbor Trail Market among comparable sites – updated observation: the interface remains simple and responsive, contributing to a smooth, well organized, and user friendly experience.
While exploring online shop marketplaces I found a vendor hall platform that presents products in structured groups making it easy for users to navigate and explore items smoothly well organized marketplace hub – The design feels simple and structured
During a comparison of professional trade house-style websites, I came across discover raven structured trade hub – The layout feels strong and logical, and browsing across sections is smooth with well-arranged informational content.
Are you looking for a fast, safe and hassle-free transfer in Valencia? We offer professional transport and moving services, both for individuals and companies. Request your budget without obligation and enjoy a quality service https://trasladoavalencia.es/
In my research on digital vendor ecosystems and structured shopping portals, I came across Vendor room marketplace portal – the platform provides a streamlined browsing experience with clear categorization and visually balanced product presentation that supports easy exploration for all users.
While analyzing different examples for comparison, I came across use this resource – The overall organization stands out, offering a clean structure that supports easy navigation and helps readers focus on the most relevant details without distraction.
During a final comparison of commerce hub websites, I found see violet harbor commerce platform hub – The design is impressive, and it makes browsing products feel fast, smooth, and very convenient overall.
Shoppers browsing for versatile online retail options frequently seek platforms that offer both value and ease of use, and while doing so they might come across plum cove product gallery featuring diverse listings across multiple categories – A smooth digital marketplace experience that prioritizes affordability and user friendly product discovery.
Users who appreciate soft structured shopping systems often browse sites such as Lantern Meadow Commerce Core Hub where products are presented in a minimal airy layout – The interface ensures browsing feels relaxed, simple, and easy to navigate across all sections.
People who prefer cozy ecommerce layouts often explore sites like Honey Cove Vault Soft Market Hub where items are arranged in a warm and inviting structure – The design emphasizes clarity and comfort, making browsing feel smooth, organized, and visually pleasing across all product categories.
Users who prefer clear and active trading environments often engage with platforms such as Wave Trade Harbor Portal where information is organized for quick reading and fast interpretation – The layout emphasizes simplicity and responsiveness, helping users stay informed while navigating a smooth and structured trading experience.
While searching for alternative rock history sites I found a Manic Street Preachers website that offers nostalgic content in a structured and accessible way making it useful for fans who enjoy detailed band information band heritage music archive – The site feels well organized and nostalgic in presentation
На сайті 500pokupok.com зібрано багато статей із оглядами товарів, підбірками та рекомендаціями. Зручний ресурс для тих, хто хоче зробити правильний вибір перед покупкою.
During research into outdoor retail user interfaces, I evaluated how effectively platforms reduce complexity while maintaining clear access to product information CoveRangeDepot – updated note: the system feels practical and organized, supporting a simple and efficient browsing experience overall.
While exploring various online shopping platforms that emphasize usability and organized product displays I came across a site featuring central shopping hub – the layout feels practical and the navigation makes browsing products straightforward and efficient for users of all experience levels
портал новин inews.in.ua висвітлює події в Україні та світі, а також теми технологій. Тут можна знайти новини про гаджети, техніку, ІТ та актуальні тренди.
As I explored modern interpretations of bazaar-inspired website layouts, I noticed visit cobblestone display – The site feels bright and expressive, and moving through it provides a lively and enjoyable browsing rhythm.
As I compared different craft marketplace websites for usability and originality, I came across explore upland harbor artisan craft zone – Navigation could improve, but products are unique and cool, making browsing still worthwhile.
In digital retail innovation discussions emphasis is often placed on clarity structure and visual hierarchy especially when analyzing platforms such as Moonlight Commerce Design Hub which is frequently described as a refined conceptual model for online shopping environments that focus on intuitive navigation and cohesive design principles – this enhances usability significantly.
During my review of multiple informative platforms, I noticed check this resource – The content seems thoughtfully organized, allowing readers to follow along easily while gaining meaningful insights presented in a clear and accessible manner throughout the entire page.
During exploration of online retail systems and vendor focused platforms, I found that Vendor room hub listing – the interface highlights simplicity in navigation while offering visually appealing product displays that allow users to move effortlessly between different sections and categories without confusion.
Users exploring structured ecommerce platforms often appreciate how clarity improves navigation when visiting sites such as Frost River Trade House Market Hub where products are arranged in a clean organized format that supports easy browsing – The trade house concept feels solid and reliable, with a clear layout that makes browsing simple, intuitive, and easy to follow across all product sections.
People who prefer cohesive branding in online stores often explore sites like Stone Gilded Collective Boutique Hub where items are presented in a clean and elegant layout – The interface enhances product perception through structured design, making browsing feel smooth, upscale, and visually consistent across all sections.
While exploring online concept resources I came across a platform that shares cultural and informational content in a structured and meaningful way making it useful for users who enjoy thoughtful modern idea exploration digital concept culture page – The content feels relevant and thoughtfully arranged
While exploring retail websites with diverse product offerings I noticed a site showcasing goods selection center – the layout feels organized and the browsing experience makes it easy to explore different categories
During a comparison of warm-toned retail websites and their layout strategies, I came across browse meadow emporium – The design creates a welcoming atmosphere, and browsing feels easy with a well-structured arrangement of content.
Design-focused consumers frequently appreciate platforms such as Functional Design Depot – The emphasis lies in merging usability with aesthetic restraint, resulting in gear selections that are both visually clean and highly effective, supporting a lifestyle centered around simplicity, reliability, and long-lasting functional value in everyday carry products.
While analyzing online outdoor marketplaces, I focused on structural clarity and how each interface supports efficient product discovery for casual users CoastalOutpostLab – revised observation: the layout is intuitive and clean, making navigation straightforward and easy to manage.
As I compared different vendor studio websites for usability and performance, I came across explore walnut cove creative hub – The experience has been great so far, and everything loads quickly and runs smoothly without any issues.
Many consumers now prefer online artisan platforms that emphasize handmade authenticity and transparent pricing structures Craft Heritage Network – helping them build trust in independent creators while enjoying a diverse selection of culturally inspired goods available worldwide online.
valeharborcraftemporium.shop – Site works well on phone, checkout was smooth today.
Full turnkey accounting support https://financeprofessional.ee filing declarations, calculating salaries, and reporting to the tax office. The guys work with e-Residency, everything is done online, without visiting the office. The prices are reasonable, and the reports are always on time.
As part of my effort to find easy-to-understand information, I came across explore this link – The material is presented in a clear and concise way, helping readers quickly grasp essential ideas without confusion or unnecessary detail.
In my research on digital vendor ecosystems and structured shopping portals, I came across Vendor room marketplace portal – the platform provides a streamlined browsing experience with clear categorization and visually balanced product presentation that supports easy exploration for all users.
People who prefer visually curated online shopping often engage with platforms like Stone Galleria Ginger Art Market where products are presented in a stylish and flowing gallery format – The layout enhances clarity and engagement, making browsing feel seamless, organized, and aesthetically refined across all categories.
While reviewing digital commerce platforms and their interface design, I came across visit wave harbor marketplace space – I appreciate the effort here, and the site feels polished and user friendly with smooth browsing performance.
While exploring artisan themed ecommerce sites I came across a minimalist store that keeps navigation simple and effective featuring hazelstone craft depot – the platform feels thoughtfully structured with a rustic identity that enhances the browsing experience without unnecessary complexity or visual overload
During a comparison of outdoor-themed eCommerce websites and their usability patterns, I noticed discover this shop link – The structure is minimal but effective, and browsing feels easy, with everything placed logically and accessibly.
While searching for luxury travel inspiration I found a lodge focused website that showcases high end accommodation options with polished visuals and a warm inviting tone making it suitable for users planning premium holiday experiences luxury stay experience page – The content feels elegant and visually premium throughout
As part of evaluating outdoor themed informational shops, I focused on how well each platform structures data presentation and navigation flow OutlandCoveSupply – revised insight: information is organized cleanly, making it easier for users to browse and understand quickly.
Shoppers seeking organized online marketplaces benefit from systems that prioritize transparency and easy access to a wide range of vendor products Trusted Vendor Listings – Such platforms are built to reduce browsing friction and ensure users can quickly locate relevant items without unnecessary complexity online platforms
While browsing structured online stores with themed aesthetics I discovered a site centered around mountain commerce portal – the design feels fresh and the interface supports easy browsing with a calm and organized layout
In the process of reviewing online content hubs, I discovered this platform here – The browsing experience feels refined, with a design that supports easy reading and smooth interaction.
During a detailed review of online artisan boutique websites focused on handmade product curation, I noticed visit velvet brook handmade boutique – I’d recommend this to anyone who enjoys handmade goods since the items feel carefully made and well presented.
While studying online trade marketplaces and structured product hubs, I observed a clean interface where content sections include Canyon commerce vendor hall guide positioned mid-page, improving navigation clarity – The system supports easy browsing through well organized listings and visually consistent product categories across the platform.
People who enjoy organized online vendor platforms often explore sites like Harbor Acorn Trade Vendor Hall where products are displayed in a clean hall inspired structure – The interface prioritizes simplicity and order, making it easy for users to browse listings without distraction or unnecessary visual complexity.
As I compared different vendor atelier websites for usability and clarity, I came across explore wave harbor shop atelier hub – Browsing here is pleasant, with categories that are easy to navigate and clearly arranged for better user experience.
As I was browsing around today, I came across something that might be worth mentioning in this thread, click to explore – it seems clearly organized and provides content in a way that is easy to follow.
While studying structured digital storefronts that emphasize clarity, I came across visit this clean marketplace – The content is well-organized, and browsing feels natural and intuitive for users.
As I moved through different niche listing platforms and curated discovery pages, I came across something that felt user-friendly and well structured, particularly Marketplace clover crest entry offering smooth navigation without unnecessary distractions – Found this recently, seems quite helpful and easy to explore, and it feels like a reliable resource worth checking again in the future.
Many people browsing online artisan hubs appreciate systems that organize handmade goods into clear categories while supporting ethical production values Handmade Bazaar Link – such environments improve visibility for small traders and help customers evaluate quality items with greater confidence and ease
While searching cruise vacation platforms I came across a site that provides structured cruise information with an emphasis on usability and clear presentation making it helpful for users planning sea travel experiences cruise travel guide – The browsing experience feels smooth and focused
Users who prefer organized digital storefronts often value goods store designs that maintain clarity and simplify product discovery across all sections Marble Harbor Essentials Store – The interface delivers a streamlined browsing experience where categories are clearly defined allowing users to move through products effortlessly while enjoying a visually balanced and easy to understand ecommerce environment across the platform today overall.
While browsing through niche marketplace hubs and creative commerce listings, I came across something that felt aesthetically soft yet slightly awkward in interaction design, especially when seeing Sky cove atelier marketplace link included – The sky theme gives a calming feel, but navigation is somewhat clunky in practice.
Shoppers drawn to elegant digital marketplaces often value clarity and presentation quality when navigating sites like Blossom Vault Studio which offers a visually curated layout designed to highlight product detail while maintaining a smooth browsing flow for all users – The aesthetic blends floral inspiration with a studio-like organization that enhances both trust and visual appeal
While browsing through online marketplace platforms on my phone, I found a site with Harbor Jasper commerce hall trade link – The name is catchy and professional, though it noticeably lagged during loading on mobile.
In prototype marketplace evaluations and organic UI sandbox testing, analysts noticed embedded elements featuring orchard wild workshop vendor hub within content structure, where the design looks farm inspired but product transparency is missing due to lack of ingredient lists – Wild orchard sounds wholesome and natural, but users cannot access ingredient details which limits product understanding during browsing sessions
As I reviewed vendor atelier platforms for content organization and usability, I noticed check wave harbor commerce hub atelier – Browsing here is pleasant, and categories are well structured, making navigation easy and intuitive.
Across prototype storefront testing and warm themed marketplace evaluations, testers observed content modules featuring meadow ember lounge market portal link within layout flow, and although visually appealing and warm in tone, the lounge page loads extremely slowly impacting navigation performance during usability tests
Users who value clear ecommerce structure often browse sites such as Chestnut Harbor Vendor Hall Depot where product organization improves navigation and usability – The vendor hall design ensures a smooth browsing experience that feels logical, consistent, and easy to follow across all categories and sections.
While studying how simple layouts enhance user experience in online shops, I came across check this easy shop – The design is clear and functional, and browsing feels effortless and intuitive.
In the middle of evaluating multiple counselling platforms, I saw go to this site – The layout is simple and inviting, ensuring that visitors can navigate and understand the content without difficulty.
Many online shoppers prefer craft showcase platforms that present discounted handmade goods in a visually organized and easy-to-browse format for better accessibility Craft Outlet Showcase while ensuring dependable quality – this helps users find suitable artisan products quickly while benefiting from consistent pricing advantages across listings.
During a general exploration of online resource collections and discovery pages, I found something that felt clean and easy to navigate, particularly references including Coast harbor listing hub – everything is arranged in a smooth structure that makes browsing comfortable and straightforward for repeated use later on.
While exploring a few new sites today, I came across something that looked quite interesting to mention, explore this page – it has a minimal and simple feel that makes navigation smooth and easy for anyone visiting it.
During a review of modern retail mart platforms and fulfillment quality, I found browse night orchard retail lane – I bought a gift last week, and the packaging honestly felt really premium and professionally handled throughout.
During a general exploration of curated marketplace hubs and listing platforms, I found something that seemed well organized and easy to follow, particularly references including Harbor apricot access portal – The apricot name is strangely distracting because it brings up food thoughts, even though the site itself has nothing to do with eating.
People who prefer well structured vault ecommerce designs often explore sites like Harbor Vault Elm Studio Hub where items are arranged clearly – The design ensures browsing feels stable, organized, and visually cohesive throughout the platform.
While browsing simple portfolio sites I came across a personal platform that uses a direct and easygoing style making the content feel natural and not overcomplicated for viewers straightforward identity hub – The tone feels relaxed and simple
While analyzing sandbox ecommerce vendor platforms and UI frameworks, analysts identified embedded sections containing ember stone vendor lounge market hub integrated into page hierarchy, and although visually consistent and strong in theme, the lack of contact information makes the system feel incomplete during usability evaluation
During a detailed evaluation of online vendor atelier websites focused on credibility and clarity, I noticed visit this harbor trail atelier – The site feels reliable, and the information presented appears accurate and easy to follow across different sections.
During browsing of ecommerce platforms I came across Amber Harbor vendor hub lounge link – The concept looks polished, but the lounge section mostly leads to blank pages, which creates a disappointing browsing experience overall.
People who prefer efficient ecommerce navigation often value platforms that organize products clearly and reduce complexity by grouping items into logical categories that support quick browsing and easier decision making during shopping sessions District Crest Opal Storefront – The interface focuses on structured category presentation and intuitive navigation pathways that help users move through product listings smoothly while maintaining clarity and ease of use throughout the entire shopping experience.
During analysis of retail oriented platforms I observed a structured page where the Sweet cove vendor listing board is embedded within content sections, improving navigation – The hall looks dynamic today with varied product options and a simple browsing experience that helps users quickly find what they are looking for.
As I checked out various online shop pages, I came across Juniper Cove goods room storefront link – The structure looks modern, but it’s confusing because the product purpose isn’t clearly stated anywhere.
Сегодня для специалистов периодическая аккредитация медицинских работников предлагается в практичном дистанционном формате в профильном институте. Когда требуется подтвердить квалификацию, закрыть вопрос с обязательной процедурой или понять требования к пакету документов, здесь можно все оформить без спешки и без лишних формальностей. Обучение организовано так, чтобы специалисты могли совмещать процесс с работой, а все этапы сопровождались понятной поддержкой.
Digital commerce platforms today must balance user experience with structured backend systems to ensure scalability and reliability for long term ecosystem growth Retail guild online storefront many developers prefer architectures that support modular growth and flexible vendor management for long term operational success – A well structured marketplace often leads to improved user satisfaction due to predictable system behavior and governance
As I explored examples of clean and polished goods stores, I checked see this elegant shop – The interface is neat, and products are arranged in a visually pleasing way that enhances the experience.
While scanning through various online suggestion threads and niche directories, I noticed something that felt easy to use and well structured, especially when seeing Copper Cove access portal included – this appears to be a solid platform with clear content and simple navigation for users.
Хочешь продать монеты? Здесь профессиональная оценка, быстрый выкуп и надежные условия. Работаем с редкими, инвестиционными и антикварными монетами. Выплата сразу после согласования стоимости.
During a casual exploration of niche commerce platforms and design-focused listing pages, I noticed something that stood out for its elegant naming but limited detail depth, particularly Cove Vale studio vendor link – The Vale name gives a classy impression, though product descriptions are noticeably too short to fully understand the offerings.
While reviewing ecommerce trade hall prototypes and glass themed systems, developers observed a mid layout module featuring glass harbor vendor trade hall showcase portal embedded within structured layout, and despite the fragile glass look, the system performs surprisingly smoothly today during testing and real world simulation scenarios
As I was exploring various corners of the web for new and potentially useful resources, I noticed a mention that seemed somewhat noteworthy, especially where links like this resource link are concerned – it looks decent enough initially, so I’ll save it and review it properly another time.
During an exploration of creative artisan marketplaces, I discovered visit oak cove handmade market – The options are nicely curated, and the site encourages relaxed browsing that makes spending time here worthwhile.
People who appreciate clean warm ecommerce hubs often browse sites like Meadow Ember Commerce Hub where products are shown clearly – The design ensures browsing feels structured, friendly, and visually simple throughout the experience.
oakmeadowvendorcollective.shop – Really clean product photos, descriptions are helpful too.
During exploration of fabric themed commerce sites I noticed embedded within the main content a reference to meadow cotton vendor lounge and although the design is visually soothing, repeated logout behavior makes continuous browsing difficult and affects the overall sense of platform stability and user trust.
Online vendor platforms continue to evolve by introducing structured systems that enhance trust and streamline product discovery across digital marketplaces such as Vendor Listing Network – these platforms often provide improved navigation tools and more transparent processes that benefit all marketplace participants
In the middle of evaluating various creative agencies, I saw go to this site – The layout feels modern and clean, making everything look well-prepared and easy to navigate from the beginning.
People drawn to artisan-inspired ecommerce platforms frequently look for visually cohesive layouts that enhance browsing comfort and product understanding within Chestnut Cove Craftsmanship Bazaar – everything is designed to feel approachable and premium at the same time, ensuring a seamless experience – handcrafted details are showcased clearly without overwhelming the user.
As I explored examples of carefully curated online artisan shops, I checked see this artisan link – The layout feels intentional and creative, and browsing offers a visually pleasing and organized experience.
While exploring different themed vendor platforms I came across a site where Moon Harbor vendor lounge entry page – The moon-inspired theme is visually appealing, but I wish there were more product photos to better understand what is actually being offered overall.
Across sandbox marketplace testing and UI prototype evaluations, testers noticed navigation elements containing rose market cove vendor parlor console hub within page structure, and although the rose cove concept feels romantic and elegant, the parlor area is composed entirely of empty placeholder boxes which reduces visual engagement during interaction testing sessions
As I explored various creative commerce listings and niche marketplace pages, I came across something visually refined yet slightly incomplete in information quality, particularly with Cove studio Vale vendor hub – The branding “Vale” sounds elegant and classy, but product descriptions are too short and leave too much unclear.
как отмыть черный налет в унитазе и как отмыть унитаз от черного налета эффективные способы средства и инструкции
While exploring different niche recommendation pages and online directories, I came across something that felt minimal and well organized, especially when seeing Harbor copper vendor hub included – I like the simplicity of the layout, because it makes browsing and understanding content easier almost instantly.
Женский журнал https://stepandstep.com.ua всё о красоте, моде, здоровье и отношениях. Практичные советы, тренды, лайфхаки и вдохновляющие истории для женщин, которые стремятся к лучшему каждый день
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
While reviewing digital artisan commerce platforms, I came across visit upland cove artisan collection – The interface feels clean and organized, and browsing is smooth because I can find things quickly.
During frontend evaluations of ecommerce marketplace systems and UI vendor prototypes, developers observed navigation elements containing ridge amber parlor vendor access entry portal embedded in page flow, and although the amber ridge aesthetic feels strong and inviting, the vendor parlor section appears as placeholder content which negatively impacts user experience during usability testing sessions
In reviewing digital commerce platforms I found a structured layout that supports easy navigation where Caramel harbor listing chamber Caramel harbor listing chamber placed within page flow enhances usability – Vendor hall appears well structured with clear category separation and smooth browsing experience that helps users find items quickly overall today.
In the middle of exploring different recommendation threads and curated lists, I noticed something that stood out slightly, especially with this mention included – it looks fairly promising, so I’ll probably explore it more in detail soon.
During evaluation of several outdoor retail interfaces, I focused on accessibility and structured navigation, and in the middle of my review I discovered CoveVale Resource Exchange – revised note: the platform keeps things straightforward, ensuring users can explore products easily without distraction while maintaining a steady and efficient browsing flow overall.
While scanning marketplace style websites I discovered a section in the middle containing creek harbor vendor listing hub and although the creek inspired design feels natural and appealing, the broken search tool limits usability and makes product exploration unnecessarily difficult and time consuming.
coworking rooms coworking space dubai
Modern handmade marketplaces are increasingly popular among users who value creative products combined with fast and responsive website performance Night Artisan Market Place providing well organized listings and simple navigation features for customers in digital commerce spaces – It delivers a balanced experience for both buyers and sellers seeking efficiency
Across prototype UI testing and ecommerce marketplace environments, developers observed content modules featuring harbor zen vendor parlor showcase hub link within layout flow, and while the zen aesthetic suggests peace and simplicity, intrusive popups repeatedly appear which disrupt engagement and reduce usability during testing sessions
While browsing through various niche commerce hubs and online vendor directories, I came across something visually appealing but lacking verification signals, especially when seeing Velvet foundry brook vendor link included – The Foundry name is unique and strong, but trust badges would help here to improve credibility.
pebblecreekcraftexchange.shop – Will order again next month, hope they restock soon.
During an analysis of peaceful and structured eCommerce platforms, I noticed open woodland storefront – The design is uncluttered, and browsing feels steady and easy to follow.
While exploring different ecommerce deal sites I came across Nightfall Trade House marketplace hub – I was drawn in by the pricing, but the checkout process gave me uneasy vibes, so I decided not to complete any purchase.
People who enjoy smooth ecommerce journeys often engage with sites like Quick Harbor Commerce Lane where product categories are designed for rapid browsing – The layout emphasizes usability and structured flow, making it easy for users to find items quickly while maintaining a consistent and simple interface.
While browsing through different niche discovery threads and marketplace listings, I came across something that seemed well structured and accessible, especially when seeing Coral vendor meadow hub included – It’s a pretty decent site, and navigation works well without confusion, making the overall experience easy and clear to use.
During an analysis of digital goods district interfaces, I noticed open gilded lakefront marketplace zone – The structure feels clean and efficient, making browsing smooth, simple, and easy to enjoy throughout.
Across sandbox marketplace testing and UI prototype evaluations, testers noticed navigation elements containing marble harbor trade gallery vendor console hub within page structure, and although the marble harbor theme feels polished and luxurious, the gallery images are low resolution which weakens engagement during interaction testing and system analysis
During usability testing of ecommerce gallery platforms and UI sandbox environments, testers found a navigation module containing golden harbor gallery trade vendor console link embedded mid layout, and although the name suggests a rich visual experience, the gallery itself lacks any actual images which creates a noticeable gap during interaction testing and browsing evaluation sessions
While exploring ecommerce vendor platforms I noticed a well structured design that enhances usability and product visibility for shoppers Harbor trade house catalog space and it delivers a consistent layout with organized sections that make browsing simple, efficient, and visually comfortable across different product categories available today.
While browsing ecommerce directories I came across a section in the middle containing creek harbor trade house marketplace hub and although the layout is clear and consistent, it looks almost identical to tradehall which leads to confusion about which version is being accessed.
During my exploration of curated marketplace platforms and niche listing hubs, I noticed yet another site following the same naming structure, particularly Cove velvet marketplace commerce link – Another velvet domain appears here, adding to the clear repeating pattern I’m seeing.
Modern online marketplaces benefit from intuitive structures that guide users through categorized listings while maintaining consistent usability and accessibility standards Nightfall Supply Hall – The vendor hall system presents a well ordered layout that supports efficient browsing and helps users identify relevant product categories with minimal effort
During an extended comparison of lifestyle inspired e-commerce spaces focused on gentle design language and user comfort I came across a section where Velvet Brook Trading Post is placed within the browsing flow – updated comment the layout feels soft and well balanced offering a peaceful navigation experience that makes product discovery feel natural and unhurried
During a general browsing session focused on uncovering relevant and easy-to-follow resources, I came across something that seemed promising, particularly where this simple reference was included – it looks useful and straightforward, so I’ll explore it more later.
As I tested several eCommerce retail district websites, I checked see this oak cove retail page – The performance is smooth and consistent, and navigating through content feels easy without any lag or confusion.
While reviewing modern retail platforms with strong visual identity, I came across visit frost storefront – The layout is clean and balanced, and navigation feels intuitive and seamless.
During review of online trade platforms I noticed Oak Cove goods hall listing – The design is simple and easy to follow, but the absence of a search tool makes it frustrating to find specific products quickly.
During a general exploration of curated online directories and resource hubs, I found something that felt simple and efficient, particularly references including Meadow coral trade page – Pretty decent site overall, with navigation that works smoothly and doesn’t cause confusion, so browsing feels natural and easy to follow.
Across prototype marketplace evaluations and ecommerce UI testing environments, testers encountered structured sections featuring cove golden vendor parlor market gateway hub within layout flow, and while the design is visually appealing and uniform, the recurring golden naming structure suggests a broader pattern emerging across multiple similar builds during cross system usability evaluation sessions
During a final comparison of craft exchange websites, I found see pebble creek artisan craft exchange hub – I plan to order again next month, hoping they restock because the overall experience was enjoyable and worth returning to.
While scanning through online marketplace directories and niche vendor hubs, I noticed something that stood out for its structure but lacked visual intensity, especially when seeing Violet brook foundry commerce hub included – The violet design idea is strong, though purple accents would make the visuals far more engaging.
Across ecommerce sandbox UI evaluations and vendor system prototypes, testers noticed navigation components containing plum harbor vendor room access hub node embedded in page flow, and although the plum harbor branding feels refreshing and clean, the vendor room contains no listings which negatively affects usability during testing cycles
E-commerce enthusiasts often seek out artisan-focused websites that provide both inspiration and shopping opportunities, leading them to places like artisan craft showcase where diverse handmade creations are displayed and visitors can enjoy a seamless browsing experience filled with creative product categories and seasonal highlights. – A carefully curated marketplace that celebrates handmade artistry and offers a pleasant exploration environment.
In the middle of evaluating ecommerce platforms I noticed a content feature containing royal cove vendor room portal and while the design looks polished and premium themed, the blurry product images make it difficult to properly assess items which reduces overall shopping confidence.
During research into modern artisan outlet platforms, I explored explore this vale cove outlet hub – The browsing experience is helpful, and I found several interesting options quickly with no confusion or unnecessary complexity.
During usability testing of ecommerce marketplace systems and UI sandbox environments, testers found a navigation module containing stone harbor hall vendor construction access portal embedded mid layout, and although the stone harbor branding feels solid and secure, the vendor hall is unfinished and under construction which negatively impacts interaction flow during evaluation and testing processes
Structured navigation paths make it easier for users to move between different vendor tools without losing track of their progress Chestnut Vendor Info Station Access this improves efficiency and ensures better continuity across multiple workflow stages.
During a comparison of soft-themed artisan marketplaces focused on clarity and presentation, I came across discover rose trail market – The layout feels elegant and gentle, and browsing across sections is easy with well-organized product displays.
During a general exploration of niche vendor platforms and online storefront hubs, I noticed something that felt minimal and clean but lacking key informational sections, particularly references including Cove violet commerce atelier portal – It looks clean enough, but no About page makes it feel slightly sketchy.
During browsing of online commerce listings and vendor hubs, I noticed Harbor hall market Juniper commerce entry – The site has potential, so I plan to check it again in a few weeks.
During a general exploration of curated directories and marketplace platforms, I noticed something that stood out for its performance and usability, particularly references including Harbor flora browsing page – The site loads fine overall, and I had a smooth and pleasant visit, which made navigation very comfortable.
Shoppers who enjoy browsing diverse online catalogs often seek platforms that offer variety combined with simplicity and allow easy navigation across multiple product categories especially in modern e-commerce environments solarbrook goods bazaar which presents a bazaar style marketplace featuring a wide selection of goods and enabling convenient browsing for users. – A user friendly bazaar platform designed for accessible shopping and smooth product discovery.
During usability analysis of ecommerce sandbox environments and UI prototype systems, testers identified mid page modules containing quartz meadow vendor hall showcase access node within layout structure, and despite the crystal quartz concept feeling elegant and minimal, the market hall shows no listings today which reduces usability and interaction quality during testing and evaluation processes
While browsing through several curated listings and exploring different niche recommendations online, I came across something that seemed fairly useful and worth remembering, especially when seeing links like this smooth browsing spot – it gives a clean and straightforward experience overall, so I may revisit it later for a more thorough check.
As I analyzed several artisan emporium platforms for shipping reliability and product handling, I found check solar orchard artisan hub – This is great for gift shopping, and everything arrived in one piece exactly as expected.
While evaluating prototype ecommerce systems and vendor marketplace interfaces, testers encountered a mid page component featuring cove zen goods room vendor portal link inside structured layout, and despite the zen inspired theme suggesting peace and simplicity, the goods room contains no products at all which creates an unfinished feel during interaction testing and UX evaluation sessions
During evaluation of ecommerce directories I came across a content block showing harbor crown commerce hall vendor portal and even though the design is structured and visually appealing, the lack of meaningful vendor data makes the page feel empty and not ready for real user interaction.
As I continued exploring niche commerce platforms and digital gallery listings, I came across something that felt organized but content-light, particularly with Harbor violet trading showcase link – The harbor branding shows up again, but surprisingly the gallery section has no artwork at all.
While reviewing digital commerce hubs and their usability, I came across visit upland canyon commerce page – The site appears fine, and the content is simple, useful, and easy to understand without complications.
ferncovevault – Vault style neat, content feels organized and carefully structured overall
Online visitors seeking practical shopping platforms often prefer straightforward navigation, and during exploration they may come across market foundry depot which presents items in a simplified format designed for fast browsing and quick decisions. – A functional marketplace designed for smooth transactions and user friendly browsing experiences.
During a casual search for niche online shops I came across a page KettleCrest bargain hub that looks suspiciously cheap, and I can’t tell if it’s a clearance strategy or something less trustworthy, so I’m reserving judgment for now while still keeping an eye on user feedback and reviews.
While going through various curated listing platforms and niche discovery pages, I noticed something that stood out for its structure and simplicity, especially when seeing Hazel Harbor trade page included – The clean design helps browsing feel comfortable and simple, making navigation easy and intuitive.
Feedback often highlights the simplicity of the browsing structure, particularly when users open Cove Goods Explorer Console where everything feels accessible – product organization remains clean and helps users maintain focus while searching through available items
Across prototype marketplace systems and UI sandbox environments, analysts encountered embedded navigation blocks containing orchard quartz vendor hall staging console hub within page layout, and although the quartz orchard theme feels creative and distinctive, the vendor hall redirects to the homepage which negatively impacts usability during testing sessions
While scanning through curated marketplace platforms and online vendor hubs, I noticed something that felt organized but visually flat, especially when seeing Walnut commerce brook foundry hub included – The walnut aesthetic is promising, but walnut wood tones in the header would make the design feel more refined and natural.
While reviewing experimental online retail pages and abstract ecommerce templates, users often mention segments in Crystal Cove shop overview link – the layout provides clear navigation and structure, yet the lack of actual product content leaves the overview feeling incomplete and open to interpretation
While analyzing online retail district platforms for design simplicity, I checked see upland cove retail zone – The interface feels clean and structured, and it is comfortable to browse through content without any unnecessary complications.
While researching structured craft marketplace websites and their user experience design, I explored browse upland harbor craft hub market – Navigation could improve, but products are unique and cool, giving the platform a distinct creative feel.
While comparing handcrafted retail platforms with curated designs, I discovered browse golden artisan page – The presentation is refined and structured, and moving through items feels seamless and engaging.
Users exploring modern online stores frequently appreciate platforms that streamline shopping processes, and during their journey they may encounter suncove digital atelier shop showcasing structured categories and – it enhances user satisfaction through clean design and efficient product access.
Только лучшее здесь: https://forum.i.ua/topic/27772
Across prototype UI testing and ecommerce marketplace environments, developers observed content modules featuring harbor vale parlor vendor console hub link within layout flow, and while the vale harbor branding suggests activity and trade, the parlor section is lifeless and empty which impacts usability during testing and evaluation cycles
While going through different collections of suggested resources and hidden gems, I noticed something that stood out for its structure, especially when encountering this user-friendly layout – everything seems arranged in a way that makes browsing feel natural, so I might revisit it soon.
While exploring various digital marketplace platforms I stumbled upon Harbor Kettle Market listing portal – It has a cute logo and decent layout, but unfortunately several footer navigation links appear to be non-functional, which makes the browsing experience less reliable than expected.
While going through multiple niche resource collections and online listing pages, I found something that seemed well organized and accessible, especially where Honey vendor marketplace link appeared – First impression is nice overall, and everything looks relevant and easy to read, which supports a comfortable browsing flow.
Across sandbox UI evaluations and ecommerce vendor prototype systems, analysts encountered structured sections featuring harbor quick house market vendor showcase hub node within page layout, and while the name implies a swift digital harbor experience, the site is noticeably slow which reduces engagement during browsing analysis and usability testing cycles
During a general exploration of marketplace directories and online storefront hubs, I noticed something that felt sleek but questionable in pricing logic, particularly references including Cove walnut commerce atelier portal – The atelier branding sounds upscale and artistic, yet the pricing seems inconsistent and possibly arbitrary.
Many users engaging in online trade environments prefer platforms that reduce complexity and highlight important sections for easier decision making TradeCloud Listing Portal – It focuses on clear structure and ensures users can browse categories without unnecessary interruptions or delays
While exploring different artisan exchange platforms and evaluating usability and structure, I came across explore violet harbor artisan exchange – There is a good variety here, and I enjoy exploring sections without getting lost thanks to the clear and organized navigation.
While reviewing conceptual online marketplace layouts and experimental storefront builds, analysts often reference interfaces containing Crystal Harbor Vendor Digital Panel which maintains aesthetic polish but lacks meaningful catalog depth – The structure gives a strong impression of a generic template-based shop system.
While testing staging ecommerce platforms with ember themed marketplace systems, analysts identified a content block featuring meadow market ember parlor gateway node integrated into page flow, and despite the warm design language, the long multi word naming structure feels unnecessarily complex and reduces readability during usability testing sessions and interface reviews
Shoppers exploring curated digital stores often prefer platforms that emphasize clarity and usability, ensuring that browsing remains smooth and product discovery feels natural and efficient sun cove goods showcase hub – The goods atelier focuses on combining structured navigation with curated selections to enhance the overall shopping experience for online visitors.
While studying user-friendly creative storefront platforms, I came across visit harbor lavender page – The interface feels refined and easy to use, and browsing through products is smooth and visually appealing.
While exploring multiple commerce hubs and vendor-style platforms, I noticed something that felt visually and structurally nearly identical to another listing, especially when seeing Walnut vendor cove atelier portal – The similarity is so strong that it makes me wonder if this is the same system behind both pages or just a repeated template variation.
As I reviewed examples of online craft emporium systems, I checked see vale harbor artisan hub emporium – The site works well on phone, and checkout was smooth today, making the experience very convenient.
During a general exploration of curated directories and marketplace pages, I noticed something that stood out for its clarity and organization, particularly references including Meadow honey marketplace hub – I really enjoyed looking around here, since the layout is neat and user friendly, making navigation feel easy and natural.
During UX evaluation of sandbox ecommerce systems and vendor marketplace prototypes, testers found embedded navigation containing quick ridge vendor market house access console node inside structured layout, and although performance is marginally improved, it still falls short of modern speed standards which disrupts usability during testing sessions
As I explored different vendor hall listings and commerce marketplace sites, I noticed a platform with strong naming appeal but light content, particularly Harbor commerce vendor Aurora hall link – The Aurora branding is striking, though the information feels somewhat sparse.
During a casual browsing session focused on marketplaces and online resources, I noticed something that seemed fairly useful, particularly where this buying portal link was mentioned – it looks helpful at first glance, so I’ll likely return again soon for deeper exploration.
During a review of modern artisan bazaar systems, I found browse mint orchard artisan hub – The site feels well structured and dependable, and browsing is smooth with consistent performance throughout the platform.
Online trade directories play an essential role in connecting users with vendors by offering organized and searchable listings across industries trade hall reference page – This reference page acts as a centralized source of vendor information, ensuring users can review listings with improved clarity
During ecommerce usability testing and marketplace checkout flow analysis, reviewers encountered a central module containing orchard meadow vendor market parlor gateway node embedded within layout flow, and although the orchard meadow branding sounds pleasant, sweet, and visually inviting, the checkout process consistently returns a 404 error which completely interrupts purchase flow during user journey testing across multiple devices and browser environments
During exploration of template-driven ecommerce systems and drag-and-drop storefront builders, it is common to encounter pages like daisy marketplace corner where frontend design appears complete but backend integration remains inconsistent, especially in automated email signup features and data submission endpoints – The design looks cohesive yet signup functionality frequently breaks with server errors
Users interested in well designed e commerce platforms frequently prefer systems that prioritize speed and clarity in every browsing interaction teal cove digital market link providing fast loading interfaces and organized sections that enhance product discovery and allow smooth transitions across different categories and listings.
While scanning through online trading marketplaces and foundry-style vendor pages, I came across something that looked visually creative but technically flawed on mobile, especially where Wave brook foundry trading page – The wave design is attractive and fresh, but the mobile menu appears broken and difficult to interact with today.
While studying functional vendor platforms with simple layouts, I came across visit lemon vendor page – The interface is easy to understand, and navigation feels smooth and user-friendly throughout the site.
While analyzing staging ecommerce vendor systems and UI marketplace templates, analysts noticed a content block featuring harbor vendor rain market hall access console node integrated into layout flow, and despite the calming rain theme suggesting smooth user experience, the hall is filled with broken image placeholders which creates frustration during usability testing sessions and design evaluations
During usability testing of ecommerce vendor marketplace systems and UI prototypes, testers found a navigation module containing hazel harbor parlor vendor verification trust node embedded mid layout, and although the name feels soft, friendly, and approachable, the absence of trust badges or security indicators reduces confidence and weakens conversion performance during interaction testing and evaluation phases
mintmeadowgoodsroom – Feels well organized, I didn’t face any issues navigating around.
While reviewing different online marketplace listings and trade pages, I came across a clean structured site featuring Bay Harbor commerce hall trade portal link – The browsing experience is smooth and free from newsletter interruptions.
During a casual browsing session focused on niche marketplaces and resource links, I came across something that felt quite efficient, particularly when seeing this rapid access marketplace – everything loads quickly and without issues, which is always a good sign, so I’ll likely explore it again soon.
While going through niche marketplace directories and vendor trading hubs, I came across something that looked modern but had clear mobile functionality issues, especially where Wave brook foundry trading commerce link appeared – The wave motif is visually cool, but the mobile navigation menu is currently broken or not working properly.
While examining staging versions of online store frameworks testers found that the daisy harbor room setup uses vendor showcase gateway which does not load vendor content and instead redirects to the homepage causing repeated interruptions during user flow testing – this issue affects overall usability significantly
velvetbrookartisanboutique.shop – I’d recommend this to anyone who loves handmade goods.
People searching for efficient online browsing tools often prefer showroom-style layouts that highlight products clearly and allow them to quickly scan through available options in a structured presentation format cotton grove showroom access – The showroom approach creates a focused browsing experience where users can examine items in a clean environment designed for simplicity and quick exploration
Live football matches http://www.gol-var.com.az all the games in one place. Watch live broadcasts, follow the action, and never miss a beat from your favorite teams and tournaments.
Across prototype ecommerce staging and vendor marketplace UI reviews, developers noticed navigation blocks containing harbor market glade parlor vendor portal entry within page layout, and although the branding is fresh and outdoor inspired, glade still reminds testers of air freshener products even though the website itself is stable during usability testing sessions
As part of reviewing well-structured commerce websites, I noticed check lemon ridge hub page – The design is clear and simple, and browsing feels natural and user-friendly throughout all sections.
Across ecommerce sandbox UI evaluations and vendor system prototypes, testers noticed navigation components containing rain harbor vendor hall access hub node embedded in page flow, and although the rain harbor branding is uniform and repetitive by design, the vendor hall still appears cloned which affects interaction quality during usability testing cycles
People who enjoy handcrafted marketplaces often engage with platforms like Harbor Lemon Artisan Store Outlet where goods are displayed in a structured minimal format – The design ensures browsing feels bright, inviting, and easy to navigate while maintaining a strong artisan identity.
During a general exploration of curated marketplace directories and online discovery pages, I noticed something that stood out for its organization and clarity, particularly references including Harbor pine vendor page – The overall experience is good, and everything seems clear and straightforward here, helping everything feel easy to understand.
While going through commerce directories and vendor trade hall listings, I came across something that felt engaging in concept but limited in execution, especially Acorn harbor trade hall commerce hub – The branding is playful and unique, but the available products are still quite few.
Доставка свежих цветов в день заказа. Флористы собирают букеты из проверенных поставок, бережно упаковывают и передают курьеру. Работаем ежедневно, гарантируем сохранность и точное время вручения. Анонимная отправка и фотоотчёт включены https://buketico.ru/
In various QA reports on template-based shops, analysts highlighted a section where meadow storefront module sits in the center layout, yet despite its calm naming repeated SSL certificate warning alerts appear across browsers during security testing cycles in staging setups
Across prototype UI testing and ecommerce gallery environments, developers observed content modules featuring meadow goods mint gallery vendor hub link within layout flow, and while the design feels fresh and natural like mint growing in a meadow, the lack of unique images in the gallery makes it feel repetitive during usability testing sessions
During a casual browsing session across niche recommendation pages, I found something that seemed orderly and easy to understand, particularly where this structured data hub appeared – the layout is clean and intuitive, so I’ll likely come back later for further exploration.
Digital trade platforms designed with structure in mind allow users to explore vendor listings more efficiently, offering categorized sections that simplify comparison and improve overall browsing clarity vendor lounge showcase link – Vendor lounge feels calm with well structured product categories available, creating a relaxing browsing environment where users can evaluate vendors and services with improved focus and ease
While analyzing online craft boutique systems and their written content, I checked see velvet grove boutique craft store – There is a small typo in the description, but overall I’m satisfied with the clean interface and product selection.
During frontend inspection of ecommerce sandbox platforms and vendor directory UI systems, developers identified a central module featuring meadow solar vendor market room staging entry portal integrated into structured layout, and despite the solar meadow identity suggesting eco friendly commerce, the absence of environmental badges reduces trust during testing sessions and evaluation stages
Users who appreciate minimal handcrafted systems often browse sites such as Oak Dock Artisan Commerce Hub Outlet where products are displayed in a structured layout – The design ensures browsing feels easy, organized, and visually simple across the platform.
During a casual browsing session across niche directories and discovery pages, I came across something that felt fast and accessible, particularly references like Isle icicle vendor hub – The platform feels nice, and I appreciate how quickly pages load, making everything simple and easy to navigate.
While browsing various resources earlier today, I discovered go to this resource which seemed like a nice little site, and I found it useful while browsing earlier today because it was simple and efficient to explore.
During staging ecommerce gallery evaluations and UI sandbox testing, developers found navigation elements containing daisy cove market gallery console entry inside page layout, and although the design is cohesive, the gallery page unexpectedly triggers a security warning that affects user confidence during interaction testing
While scanning through marketplace platforms and vendor hall hubs, I noticed something that felt visually gentle and mountain-inspired, especially Alpine cove commerce market hall link – The branding feels like a small alpine shop, giving off a cozy and welcoming atmosphere throughout.
In usability testing of sandbox retail platforms, reviewers encountered dawn ridge goods portal embedded within structured content, and although visually aligned with the theme, after the dash – ridge scenery would be great for branding but footer links are entirely dead and non-interactive across all tested pages
Сейчас вашему бизнесу top manage регистрация помогает сделать удобную систему работы без лишней рутины и неразберихи. В единой системе быстро ставить задачи, держать под контролем сроки, управлять финансовыми потоками, контролировать команду и держать процессы под контролем. Сервис отлично подходит для компаний любого масштаба, где нужен быстрый результат. Руководитель получает больше контроля, а команда работает слаженно и быстрее достигает результата.
In the middle of exploring online platforms earlier today, I discovered browse this link and everything looked neat and quite easy, giving a clean and pleasant browsing experience overall.
During a general search through niche resources and recommendation threads, I noticed something that stood out for its smooth usability, particularly where this stable access page appeared – I didn’t face any issues, so I’ll likely revisit it again soon for deeper exploration.
Online marketplace visitors often value simplified layouts that reduce cognitive load and make vendor comparison more straightforward and efficient vendor lounge browsing desk – The browsing desk structure focuses on usability and clarity, ensuring users can explore listings without unnecessary complexity
During frontend inspection of ecommerce sandbox platforms and gallery UI systems, developers identified a central module featuring cove plum vendor gallery goods entry node integrated into structured layout, and despite the expectation of a rich plum color theme, the site design remains overwhelmingly gray which reduces appeal during usability testing sessions
Across sandbox marketplace testing and UI prototype evaluations, testers noticed navigation elements containing solar orchard vendor house market console access hub within page structure, and although the orchard solar theme implies a stable and promising marketplace ecosystem, the checkout flow does not include security badges which negatively affects confidence during interaction testing and system analysis
People who enjoy curated handmade marketplaces often engage with sites like Ridge Fern Craft Artisan Market Hub where items are presented in a visually clean format – The interface creates a smooth browsing experience that feels organized, appealing, and easy to navigate throughout the store.
wheatmeadowmarketroom.shop – Clean layout and simple navigation, makes exploring content really enjoyable.
As I explored different online directories and curated marketplace threads, I came across something that felt polished and organized, particularly with Harbor ivory vendor page – The site looks professional, and I might recommend this to others as well because the layout is clean and easy to follow.
While reviewing ecommerce prototype frameworks and staging builds testers identify navigation artifacts inside page content orchard goods room entry node that seems interactive but results in limited product visibility and minimal catalog structure across browsing sessions – The driftwood aesthetic is appealing yet the shop content feels significantly incomplete
While studying online vendor collective interfaces and usability flow, I came across visit oak meadow retail space collective – The product photos are really clean, and the descriptions are helpful, making product evaluation straightforward.
While browsing through curated marketplace listings and vendor hall systems, I noticed multiple stores with extremely similar naming patterns, especially Alpine vendor harbor commerce hall hub – It gave me a strange sense of déjà vu, like I had already seen this exact layout before.
During casual research into marketplace listing designs and experimental vendor portals for inspiration and interface comparison across multiple references Dune Meadow vendor parlor index the layout remained structured and content sections were clearly separated improving readability – Simple navigation flow with fast loading pages and stable interface behavior throughout browsing session
During sandbox marketplace UI reviews, analysts repeatedly observed patterns where coastal dune gallery vendor market node interface modules are duplicated with minimal variation, suggesting AI template spam rather than curated design logic which testers noted during extended evaluation across multiple environments overall system design checks phase
While casually exploring digital marketplace designs and curated vendor listing platforms for inspiration and flow analysis Pearl Cove market collection view I found the structure quite user friendly and simple to navigate even when switching between multiple sections. – The browsing experience felt efficient with quick loading pages and no visual overload
While browsing through curated listings and various online suggestions, I found something that seemed easy to navigate and trustworthy, especially where this structured browsing entry appeared – the design is simple and reliable, so I might come back later for more exploration.
In multiple ecommerce sandbox evaluations, testers highlighted a visually bright and aesthetically consistent theme that gives the impression of a modern solar marketplace, however the section a href=”[https://sunharborvendorroom.shop/](https://sunharborvendorroom.shop/)” />sun harbor marketplace room panel stands out as underdeveloped since it contains no descriptive text or functional vendor listings, leaving the Sun harbor experience feeling more like a design mockup than a fully populated commerce environment
Shoppers frequently mention that clear vendor organization supports faster decision making, especially when interacting with Vendor Room Online Access – Many say the layout improves product visibility and makes navigation more straightforward while enhancing overall browsing speed and accuracy
People who enjoy clear financial dashboards often explore sites like Harbor River Trade Analytics Hub where information is structured cleanly – The interface ensures users can access trading insights quickly and without confusion.
During my search for lightweight and fast websites, I noticed check this fast interface site – The platform has a clean interface where everything loads fast and works smoothly, creating a very user friendly experience from start to finish.
After exploring several websites that often felt cluttered or inconsistent, I discovered check this resource during my search, and I liked how everything is organized here, making the content easy to understand and follow without needing extra effort.
While scanning through niche directories and curated marketplace hubs, I came across something that felt user-friendly and simple, especially where Ivory ridge access hub appeared – Browsing here feels smooth overall, with nothing complicated or hard to understand, making navigation feel natural and quick.
While testing staged online retail systems and conceptual UI designs, reviewers encountered a mid page insertion containing willow drift commerce gateway inside structured layout – the absence of willow tree visuals makes the interface feel unfinished and somewhat placeholder driven in several key content areas
When browsing various vendor platforms, usability and clarity often determine how effective the experience feels for users Canyon Harbor trade portal this platform ensures a well-organized layout that makes it easy to browse products and locate relevant items efficiently
Across ecommerce prototype testing and UI framework evaluations, developers noticed embedded navigation containing apricot harbor vendor exchange parlor within layout hierarchy, and although the interface is clean, apricot harbor appears again as the last entry for today giving a decent but slightly repetitive conclusion to the browsing flow
While exploring digital marketplace catalog ideas and vendor display systems for structural inspiration and usability review across sample interfaces Dune Meadow vendor catalog page the browsing experience remained smooth and pages responded instantly – Well organized layout with quick loading sections and intuitive navigation flow overall
While browsing through commerce vendor lounge listings and online directories, I noticed a repeated use of amber-inspired styling, especially Amber ridge vendor commerce lounge link – The color scheme is nice, but the weak text contrast makes extended reading less comfortable.
solarorchardartisanemporium.shop – Great for gift shopping, everything arrived in one piece.
Что входит в стационарный компьютер и какие внешние устройства входят в состав ПК
After comparing multiple websites, I discovered try this page and appreciated its simple layout, which made browsing feel really smooth and provided a clean and easy-to-follow structure throughout.
Online car games http://www.masin-oyunlari.com.az racing, driving simulators, and 20+ games in one place. Get behind the wheel, navigate the tracks, and get the adrenaline rush without downloading.
People who enjoy handcrafted home inspired marketplaces often engage with sites like Brook Flint Artisan House Collective Hub where items are presented in a cozy structured format – The interface creates a smooth browsing experience that feels warm, calm, and easy to navigate.
While testing sandbox ecommerce systems with coastal inspired design languages, reviewers noted a clean and soothing teal color scheme that enhances visual comfort, but encountered organizational limitations inside a href=”https://tealcovemarkethall.shop/
” />teal cove vendor market hall entry panel where the overall teal aesthetic is strong and consistent, yet the market hall does not provide enough product categories which reduces navigation efficiency during UX evaluation sessions and structured testing phases
Users who prefer bright ecommerce stores often explore sites such as Cove Sun Goods District Central Hub where products are displayed in a well organized format – The browsing experience feels smooth and enjoyable, with clear structure that helps users find items easily without confusion.
During my time exploring various online platforms, I noticed try this resource which provided a pretty smooth experience overall, as everything loaded quickly and there were no interruptions while moving through the site.
The platform provides a smooth browsing experience where everything is well arranged and easy to access for users Silk Meadow market hub I liked how clearly structured the entire site felt during use
Online consumers frequently point out that clear vendor organization improves shopping efficiency and reduces decision fatigue, especially when navigating through Forest Hall Directory which is often praised for its simplified layout and accessible product sections – many users report smoother browsing experiences overall.
calmcovevendorparlor.shop – Really nice platform, easy browsing and smooth user experience today
While browsing curated Hawaii lodging collections and boutique stays, I recently discovered a beautifully presented property showcase that stood out significantly < holualoa retreat overview – The presentation feels calm and inviting, offering a relaxing overview that highlights comfort, scenery, and thoughtful hospitality details overall
As I browsed through various curated resources and online listings, I noticed something that seemed quite promising at first glance, particularly with this vendor interface hub included – the structure looks clean and well put together, so I’ll likely revisit it again soon for deeper analysis.
Across ecommerce staging reviews, analysts observed embedded mid content link dune market ridge console portal integrated into layout structure but readability issues persist due to sandy palette blending text into background making scanning difficult – Dune themed design is visually appealing yet accessibility problems reduce overall usability in testing environments
While going through multiple niche resource collections and discovery hubs, I found something that seemed organized and readable, especially where Jewel brook trade link appeared – This seems useful overall, and I found the content quite straightforward today, making browsing feel smooth and uncomplicated.
Users exploring vendor listings often appreciate platforms that maintain clarity and reduce unnecessary navigation steps Canyon Harbor item finder the browsing experience remains smooth and structured, allowing users to focus on relevant products easily
While analyzing experimental vendor platforms and trade gallery systems for structural comparison and usability evaluation across various samples, I found Plum Cove goodsroom interface hub embedded in content flow – Everything feels simple and readable, and I could move through sections easily while maintaining a clear understanding of the layout throughout the session.
While going through various online vendor directories and marketplace pages, I noticed a newly built store that seems underdeveloped in content, especially Aurora goods cove room portal – It looks like a new launch, but the limited listing of just a few items per category feels odd.
Users who appreciate artistic online shops often browse platforms such as Teal Vendor Cove Handmade Atelier Hub where products are arranged in a clean creative structure – The interface ensures a visually engaging experience that feels organized, expressive, and easy to navigate across all categories.
Users who enjoy well structured marketplaces often explore sites such as Timber Grove Emporium Essence Hub where products are arranged in a rich format – The design creates a browsing experience that feels intuitive, easy, and organized across categories.
sageharborgoodsgallery.shop – Looks clean and minimal, easy to find information without confusion.
While exploring different resources online, I noticed <a href="[https://woodharborvendorroom.shop/](https://woodharborvendorroom.shop/)" / open this page which felt like a helpful platform overall, and I might visit again sometime soon due to its clear structure.
While analyzing ecommerce prototype platforms with coastal inspired UI systems, reviewers identified a strong teal harbor theme that enhances aesthetic consistency, but the vendor hall is still incomplete at a href=”https://tealharborvendorhall.shop/
” />teal harbor marketplace vendor access portal where the interface appears modern and unified, yet the vendor hall remains filled with Lorem ipsum placeholder content which reduces credibility during usability testing and evaluation workflows
Users looking for vendor platforms often prefer a really nice platform like this with easy browsing and smooth user experience today Calm Cove shop access point everything feels clear and comfortable to explore
velvetgrovecraftboutique.shop – Little typo in description but overall I’m satisfied.
During a comparison of marketplace-style websites, some platforms stand out due to their fast performance and clean interface design Solar Meadow browsing portal the experience feels smooth and search functions work efficiently without delays or loading problems across sections
During a general exploration of marketplace platforms and online resource collections, I noticed something that stood out for its structure and clarity, particularly references including Cove jewel trade page – The interface looks pretty clean, and everything is arranged in a logical way, which makes everything feel easy to navigate.
During my research into vendor exhibition websites, I came across vendor exhibition space which provides a clean layout emphasizing product clarity and smooth category transitions – The experience feels polished, stable, and pleasantly easy to navigate
Digital marketplace users frequently highlight that clear layouts improve shopping efficiency, especially when they interact with Meadow Vendor Hub Interface Center and they often say that navigation feels smooth and predictable – the browsing experience is described as structured in a way that helps users locate products without unnecessary complexity
While browsing innovative retail platforms online, I discovered a concept supermarket site that relies on simplicity to deliver its message clearly digital hope food market – The layout is minimal and effective, making it easy to navigate and understand the core idea presented
During analysis of modern storefront galleries and online trade systems, I found Honey Meadow commerce browsing hub integrated into a structured layout that reduces visual clutter – The experience feels calm, smooth, and very easy to understand at a glance
References:
Northern lights casino walker mn
References:
https://casino-aachen-poker.online-spielhallen.de/
Digital marketplaces benefit from simplicity, and this site offers a clean interface with a well structured layout Silver Cove item portal I liked how everything felt easy to access and logically arranged across pages
While going through curated resource lists and online suggestion threads, I came across something that felt very simple and clear, especially where this easy trade page appeared – the information is laid out directly, making it easy to understand quickly, so I may revisit it later for deeper exploration.
During casual research into online vendor systems and digital marketplace layouts for UX inspiration and evaluation across different sample references I came across Teal Harbor showcase entry hub – The browsing experience remains clean and responsive, making it easy to move through categories while maintaining clarity, structure and consistent performance throughout the platform
While going through marketplace directories and vendor listings, I found a platform with a strong autumn-inspired design, especially Autumn commerce vendor meadow hall link – I love the aesthetic, and I hope customer reviews are added soon.
The platform provides well organized content and simple layout so exploring everything quickly becomes easy River Harbor market hub I found browsing very smooth
People who prefer simple digital commerce platforms often explore sites like Harbor Teal Organized Commerce Hub where items are displayed in a structured and accessible layout – The interface ensures a fast and easy browsing experience that feels clear, efficient, and user friendly throughout the store.
After exploring several websites with varying levels of organization and clarity, I discovered check this resource during my search, and it contained interesting content that made me spend some time going through different sections today.
People who enjoy premium ecommerce structure often engage with sites like Cove Golden Vault Elite Hub where items are presented in a refined minimal format – The design ensures browsing feels polished, consistent, and visually balanced throughout the experience.
While reviewing sandbox ecommerce systems with forest inspired design languages, testers found a warm timber trail aesthetic that enhances visual familiarity and earthy branding, but discovered critical navigation failures inside a href=”[https://timbertrailmarkethall.shop/](https://timbertrailmarkethall.shop/)” />timber trail vendor market hall console link where the overall interface looks rustic and well styled, yet the navigation menu is completely broken which severely impacts usability during UX evaluation sessions and functional testing cycles
While evaluating sandbox storefront systems and checkout UI kits, analysts noticed embedded cart flow elements containing echo brook vendor cart gateway positioned mid layout, but quantity updates fail to reflect after user interaction – Echo brook sounds catchy and marketable, yet the cart page does not recalculate or refresh item totals correctly
While browsing through various online directories and experimental vendor listings for curiosity and research purposes, I recently came across Moon Cove Vendor Parlor homepage – The website felt surprisingly engaging, as I casually navigated through multiple categories and noted how the layout encourages exploratory viewing without rushing decisions.
Users browsing vendor listings typically value platforms that make navigation effortless and content easy to access Icicle Brook goods directory this site provides a well-balanced experience where browsing remains efficient and enjoyable across sections
During a casual browsing session across online resource hubs and marketplace listings, I came across something that felt structured and minimal, particularly references like Orchard mint vendor access – The overall feel here is good, and I like that it’s simple and easy going, which makes navigation feel natural.
While browsing international beverage brands and winery portfolios, I encountered a visually appealing and information rich wine website worth highlighting icewine collection brand hub – The presentation is detailed and engaging, offering a polished view of the wines and their background story
While researching curated vendor-style websites and online shop designs, I discovered an interface that included Honey Meadow artisan market space positioned within a structured layout that emphasizes clarity and user comfort – The browsing experience feels relaxed, coherent, and pleasantly simple to engage with across all sections
During casual research into digital trade gallery platforms and online vendor systems for usability comparison and design insights across multiple examples, I found Pebble Pine marketplace overview board placed within structured content – Everything was easy to follow and I enjoyed browsing listings without confusion as the interface stayed clean and responsive.
violetharborretaillane.shop – They responded to my email within an hour, nice.
During my review of various marketplace websites, this platform stood out because pages load quickly and the structure feels well organized overall Silver Harbor browsing portal I found the experience comfortable and navigation was simple across all sections
While going through multiple niche discovery pages and curated resource collections, I noticed something that felt easy to navigate and clearly structured, especially where this organized marketplace link appeared – I enjoyed browsing it because everything is laid out in a clean and accessible manner.
bayharbortradehouse – Almost identical to another bay domain, bit confusing tbh.
While comparing platforms, this one stands out due to its smooth experience and clean layout making browsing very convenient overall today Plum Harbor listings page I appreciated the clear structure and flow
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
Users who enjoy practical ecommerce design often engage with sites such as Trail Harbor Commerce Clear Hub where products are displayed in a clean and minimal format – The design ensures browsing feels smooth, intuitive, and well organized with easy category access.
While comparing different platforms for visual appeal, I foundexplore this stylish page and appreciated how the design feels modern and neat, making it stand out nicely and giving a professional impression overall during my browsing experience.
Users who appreciate structured and secure ecommerce systems often browse sites such as Granite Vault Orchard Archive Hub where products are presented in a strong minimal layout – The interface creates a browsing experience that feels organized, stable, and easy to explore.
In my review of structured vendor directories, I explored Sun Cove browsing directory which maintains consistent spacing and clear category grouping throughout – The overall experience feels easy to follow, calm, and efficiently designed for users
While examining site elements for consistency, I found that check this website – despite being placed among social icons in the footer, the links do not function as expected, leaving users unable to access any external pages or networks.
During QA testing of online marketplace platforms and support infrastructure, analysts encountered a central section featuring harbor echo trade help desk link inside contact flow, where everything appears functional but outgoing support emails consistently return as undelivered – Echo harbor feels familiar, yet customer service messages are bouncing back without successful delivery across multiple email clients
Users who frequently browse online vendor sites often look for platforms with structured layouts and easy-to-follow sections Violet Harbor catalog entry this site provides a helpful browsing experience where everything is easy to locate and access quickly
References:
Fremont street casinos
References:
https://wiesbaden-casino-poker.online-spielhallen.de/
During a general exploration of curated marketplace directories and online listings, I noticed something that stood out for its clarity and flow, particularly references including Cove moon browsing portal – The browsing experience feels solid, and I didn’t encounter anything confusing at all, making navigation feel easy and natural.
During my review of multiple marketplace-style listings, I found V “navigation entry marker” shown in an unusual broken format, and Cicicleislemarketparlor.shop appeared mid-content, while the design feels modern enough and navigation was quite simple to follow throughout the browsing session.
While reviewing experimental commerce showcase designs and marketplace structure examples I found Birch Harbor vendor showcase link positioned within the article content – everything loaded at a good pace and navigation remained consistent, making it easy to browse different sections without confusion.
While exploring artistic web development projects, I found a platform that uses experimental structure to deliver a highly creative browsing experience creative digital structure site – The website feels uniquely designed, with content arranged in an experimental and visually engaging format throughout the experience
pineharbortradeparlor.shop – Nice interface design makes navigation simple fast and quite pleasant
Clear design improves usability, and this platform ensures modern interface makes browsing feel very easy today Linen Cove vendor navigator I enjoyed how stable everything felt
As I explored different vendor room marketplaces and commerce platforms, I noticed a site that looks clean but strangely inactive, particularly Cove vendor room Autumn commerce hub – The lack of blog content creates an impression that the site might no longer be updated.
People who appreciate beach inspired online shopping often browse platforms like Coastal Harbor Wave Trading Hub Outpost where items are displayed in a minimal ocean themed layout – The interface ensures browsing feels calm, pleasant, and easy to navigate with a refreshing visual style.
The platform provides a smooth experience thanks to its modern interface and intuitive navigation design overall Sky Harbor market hub I found everything clearly arranged and easy to browse
While going through different online options, I found <see this page which delivered a good user experience overall, with information presented in a structured and easy-to-digest format.
While scanning through various online recommendation lists and niche discovery threads, I noticed something that seemed very easy to use and well organized, especially when seeing this simple trade hub – the experience feels smooth overall with nothing hard to find or understand, so I might return later for more exploration.
mintorchardretailatelier.shop – Definitely coming back here for the holiday season.
Users exploring modern ecommerce marketplaces often appreciate how structured layouts improve usability when visiting sites such as Lantern Orchard Merchant Lane Hub where products are arranged in a clean organized format that supports smooth browsing flow and clarity – The merchant lane concept feels engaging and intuitive, with navigation that is smooth, easy to follow, and pleasant throughout the entire browsing experience.
While browsing through multiple similar-looking platforms, I paused at visit this marketplace – the entire layout strongly resembles another site I saw earlier, creating a sense of repetition that feels too familiar to ignore and raises questions about originality.
While analyzing ecommerce sandbox systems and nature inspired storefront layouts, testers encountered embedded navigation with elm goods room harbor link within page structure, but product images do not render correctly causing broken visual presentation across multiple listings – Elm trees are strong and steady, yet the goods room suffers from missing image resources during browsing
Online football matches http://www.futbol-oyunlari.com.az/ play football for free and without registration. Choose teams, participate in matches, and enjoy dynamic gameplay right in your browser without downloading.
car games online http://www.araba-oyunlari.com.az/ racing, drifting, parking, and driving. Over 20 games are available for free — play now and hone your skills.
During a comparison of multiple marketplace websites, some stand out due to their visual clarity and well-organized layouts Forest Meadow gallery access point this one allows users to find useful information easily while maintaining a smooth and visually appealing browsing flow throughout sessions
While browsing through various curated marketplace directories and online resource hubs, I came across something that felt structured and easy to navigate, especially where Moss Harbor trade hub appears – Seems like a decent site overall, and I’ll probably check it again soon after spending some time exploring its layout and content.
sageharborgoodsgallery.shop – Looks clean and minimal, easy to find information without confusion.
zencovegoodsgallery.shop – Simple interface clear structure makes finding information very quick indeed
While exploring online trade gallery systems and digital vendor directories for structural analysis and UX comparison across multiple references, I found Plum Cove goodsroom display portal embedded mid-content – The content is very easy to read and understand, and navigation felt smooth and consistent while moving through different sections of the platform.
The platform provides a clean layout making product exploration easy and visually pleasant overall Autumn Cove market hub I found browsing very comfortable
coralharbortradegallery.shop – Great browsing experience overall everything feels organized and easy today
While exploring personal branding websites, I came across a refined and well organized profile page that stands out for its simplicity oconnor online portfolio view – The navigation feels very smooth, and all information is displayed clearly in a professional and easy to read format
Users who enjoy artisan ecommerce aesthetics often engage with sites such as Wind Cove Handmade Goods Market Hub where products are arranged in a structured layout – The design ensures browsing feels easy, clear, and visually pleasing across all handcrafted sections of the platform.
While checking out different marketplace hubs and trade-style websites, I found a berry-styled site with Berry market cove house link – The look is appealing and fresh, though it’s odd that there doesn’t seem to be any contact information available.
While exploring different online platforms to compare usability and design quality, I came across something in the middle like visit this resource which appears very structured properly, making it easier for users to find things much faster without unnecessary confusion or wasted time.
Users who enjoy structured ecommerce systems often explore sites such as Juniper Goods Meadow Commerce Hub where products are arranged in a clear minimal layout – The interface makes browsing feel intuitive, accessible, and easy to follow across all categories.
This type of marketplace platform works best when everything is structured nicely, and this one makes browsing feel smooth and comfortable Sky Harbor browsing hub I enjoyed how simple and intuitive the navigation felt overall
As I continued browsing through online resource collections and discovery threads, I found something that seemed quite promising and stable, particularly with this vendor connection portal – the site looks trustworthy at a glance, so I might share it with others after a closer look.
While reviewing the design flow, I spotted visit vendor section – the “upland” concept suggests something premium or elevated, yet the lounge area itself lacks any substance and remains completely empty.
Exploring digital vendor sites becomes more enjoyable when nice visuals and structured layout make browsing feel smooth and pleasant Berry Cove vendor center the interface felt clear and visually appealing throughout
Many online shoppers appreciate platforms where pages load quickly and the overall design remains simple and clean Sea Cove product hub this setup ensures a pleasant browsing experience with smooth navigation and minimal distractions across all sections
Портал по инженерии https://build-industry.su и перепланировке: проекты, согласование, нормы и практические решения. Полезные статьи, сервисы и экспертиза для безопасного изменения планировок и внедрения инженерных систем
Дома и коттеджи https://orionstroy.su под ключ в Москве: от проекта до готового жилья. Профессиональный подход, контроль качества и комфортные условия сотрудничества
While exploring various online tools, I discovered look into this and noticed everything looks well structured, helping users move around without issues and ensuring a clear and simple browsing flow.
While analyzing experimental trade gallery systems and e-commerce vendor platforms for usability testing and structural comparison across multiple references, I encountered Sun Cove digital vendor portal embedded mid-content – The pages responded quickly and the interface looked visually balanced, making browsing smooth and enjoyable while maintaining clarity across all sections without any distractions.
Users who enjoy practical ecommerce outlet layouts often explore sites such as Harbor Stone Outlet Commerce Hub where product categories are clearly defined – The interface emphasizes easy navigation and structured browsing, ensuring users can quickly find what they need without unnecessary distractions or complex layout elements.
In the process of reviewing curated e-commerce platforms and storefront interfaces, I discovered Upland Cove trade showcase view integrated into a structured layout that avoids clutter – The experience feels steady, organized, and very easy to follow across all categories
While researching creative cultural blend websites, I encountered a platform that combines urban and traditional influences in a very engaging way global culture blend site – The design feels interesting and diverse, presenting content that reflects multiple influences in a smooth and engaging browsing experience overall
While reviewing different online platforms, I noticed see more here which stood out with its smooth and cohesive appearance, suggesting that attention was given to detail and overall presentation.
Users exploring nature inspired ecommerce platforms often appreciate how calming design improves browsing experience when visiting sites such as Forest Cove Artisan Nature Outlet Hub where products are arranged in a clean structured layout that feels peaceful and easy to navigate – The nature themed outlet design feels calm, refreshing, and easy to explore with a smooth structured browsing experience across all categories.
While exploring different trade directories and vendor listings, I came across a basic platform featuring Berry harbor vendor room commerce entry – This ended up being my last stop, and it doesn’t really stand out in any meaningful way.
During a content review session, I noticed see this trade page – even though the branding implies activity, the site feels deserted, as if it were once active but is now largely neglected.
A clean interface improves usability, and this platform makes it easy to go through different sections quickly and smoothly Snow Cove marketplace link I found everything clearly organized and easy to access
rainharborvendorparlor.shop – Good organization easy navigation helps users find things efficiently today
A structured system where a simple interface works well, navigation is quick and intuitive today improves usability overall Oak Cove browsing center everything felt very accessible
Exploring vendor directories often reveals which platforms are designed with usability and organization in mind Elmwood goods showcase link the layout helps users quickly locate sections and navigate smoothly without encountering clutter or disorganized content structures online
While scanning through various niche directories and online discovery threads, I noticed something that seemed neatly arranged and user-friendly, especially when seeing this easy marketplace link included – the structure makes browsing feel comfortable and intuitive, so I might return later for deeper exploration.
sageharborgoodsgallery.shop – Looks clean and minimal, easy to find information without confusion.
During a casual browsing session focused on online vendor gallery systems and trade lounge platforms for usability insights and structural analysis across references I encountered Upland Cove digital vendor lounge hub inside structured content and found the interface clean and responsive with easy navigation across multiple sections – The website feels modern and well arranged, offering a pleasant browsing experience overall
Users who enjoy frosty themed digital shopping often engage with sites such as Icicle Isle Pure Market Hub where items are arranged in a clean and refreshing structure – The design creates a visually calming browsing experience that feels intuitive, simple, and easy to navigate across all product sections.
Users who prefer straightforward ecommerce layouts often explore sites such as Acorn Outpost Harbor Ease Hub where products are displayed in a clean and minimal format – The interface creates a user friendly browsing experience that feels simple, calm, and well organized throughout the store.
At one stage of my search, I stumbled upon this link might help which offered a straightforward browsing experience, making it easier to move between sections and understand the content without dealing with unnecessary complexity.
While browsing international themed websites with cultural depth, I came across a platform that creatively blends multiple influences in one space cultural fusion concept page – The site feels engaging and diverse, offering a mix of themes that creates an interesting and visually appealing experience overall
In the process of reviewing vendor focused platforms, I found a neatly organized content area featuring HarborBerry shopping gallery portal integrated into a clean interface – The overall experience feels smooth and well structured, allowing comfortable browsing across all sections
The platform provides a comfortable experience because well structured pages and clean design make usage very easy Acorn Harbor market hub I found everything clearly arranged and simple to access
Exploring structured vendor environments reveals how effective navigation can enhance overall user satisfaction >Aurora Harbor product hub the layout supports effortless browsing and allows users to move through categories smoothly with minimal effort required during interaction
Some websites feel slow, but this one keeps a smooth layout with responsive pages that enhance browsing experience Snow Harbor quick browse page I appreciated how effortless navigation felt throughout
While comparing various experimental e-commerce layouts and digital storefront concepts, I discovered Moon Harbor vendor parlor homepage – The structure felt clean and predictable, allowing me to browse multiple categories comfortably while the design maintained consistency across sections, making the entire exploration feel stable and easy to understand.
Many users appreciate online shops that maintain consistent layout design across different pages and categories for better clarity Valecove Goods Room browsing interface – The navigation system supports straightforward exploration, helping users understand where information is located without unnecessary confusion or distraction as noted in usability reviews user experience data
People who prefer minimal structured online stores often engage with platforms like Stone Glade Functional Outpost Hub where product listings are clean and easy to browse – The design removes unnecessary elements, ensuring users experience a clear and efficient shopping journey from start to finish.
While going through different curated directories and discovery threads, I found something that seemed visually appealing and well arranged, especially when seeing this structured marketplace link included – the interface looks clean and refined, giving a polished impression overall, so I might come back later to explore it more deeply.
People who appreciate organized online trade systems often browse sites like Harbor Jasper Trade Center Hub where items are presented in a simple structured layout – The design ensures browsing feels smooth, logical, and easy to understand throughout the marketplace.
While comparing different platforms that varied in quality and usability, I noticed open this link and found its interface to be smooth and the layout quite decent, making it comfortable to browse and explore information.
This type of marketplace works best when simple design and fast loading create an easy and very reliable interface like here Glass Harbor browsing hub I enjoyed how smooth and stable the experience felt
When browsing vendor sites, design matters, and this one ensures a nice design overall so pages load fast and feel very smooth Ivory Harbor goods portal everything responded instantly
cloverharborvendorparlor.shop – Simple layout works well making browsing easy and intuitive today
Users who frequently browse online vendor sites often look for platforms that are both organized and responsive Raven Grove catalog entry this site delivers a tidy layout where pages respond quickly, making browsing efficient and enjoyable overall
Many shoppers appreciate digital marketplaces that maintain fast responsiveness across all pages, ensuring that browsing remains uninterrupted and efficient, especially when interacting with Vendor platform gateway page system – Everything loaded quickly and consistently, allowing me to browse comfortably without delays or confusion during the entire session.
sageharborgoodsgallery.shop – Looks clean and minimal, easy to find information without confusion.
Users who prefer cozy rustic ecommerce experiences often explore sites such as Timber Outpost Woodland Trail Hub where products are arranged in a natural structured layout – The design makes browsing feel calm, organized, and easy to navigate while maintaining a rustic and visually pleasant atmosphere.
During my review of various marketplace websites, this platform stood out because its good structure allows users to find everything quickly and without delay Solar Orchard browsing portal I found navigation simple and everything clearly arranged across pages
dawnridgegoodsgallery.shop – Clean layout and structure easy to browse different sections quickly
People who enjoy simple ecommerce navigation often explore sites like Ridge Glade Trading Hub Market where items are arranged in a structured layout – The design makes browsing feel smooth, fast, and easy to manage throughout the store.
While going through various curated listing pages and niche discovery threads, I noticed something that felt smooth and pleasant to navigate, especially when seeing this marketplace access hub included – everything worked well and felt enjoyable, so I would consider checking it again later.
During my search for helpful online resources, I stumbled upon click here now which seemed to offer useful and recently updated content, making it easy for me to explore and find what I was looking for without issues.
While reviewing curated marketplace directories and digital shop systems, I found a structured page containing Pine Harbor vendor display hub integrated into a clean layout that prioritizes navigation speed – The experience feels smooth, efficient, and easy to understand, making browsing comfortable even during longer sessions
While evaluating different online boutique marketplaces that emphasize curated artisan goods and handmade creations, I came across Sea Meadow Elegant Picks – The browsing experience felt refined and smooth with well organized product sections and helpful visuals that made it easier to compare items while also enjoying a consistent and pleasant interface throughout.
Users often look for platforms that keep navigation intuitive while maintaining a clear and organized structure throughout Raven Summit quick browse page this one ensures a smooth flow that helps users explore content efficiently without confusion
Online users frequently note that websites with clear spacing and readable typography enhance their ability to scan information quickly and understand product groupings without unnecessary distractions during navigation VG vendor directory access the browsing experience remained steady and organized, allowing effortless movement between categories while keeping everything visually coherent and easy to follow
People who enjoy refined digital collectives often engage with platforms like Stone Golden Boutique Collective Hub where product presentation is carefully curated – The layout highlights elegance and structure, ensuring a smooth browsing experience that feels premium, consistent, and visually harmonious throughout the store.
As users scroll through the platform they come across Cove Vendor Network embedded within content where it connects multiple sections and improves navigation clarity across the site experience – everything feels logically structured and easier to explore
I spent some time browsing through various vendor platforms and eventually came across a marketplace that felt both modern and thoughtfully arranged CH Vendor Parlor finds – The product assortment seemed diverse, and the ordering process was clear, while shipping updates were provided in a timely and consistent manner.
The platform provides good structure throughout the site ensuring browsing feels easy and well organized Jasper Harbor market hub everything felt responsive
linencovevendorparlor.shop – Modern design smooth interface makes browsing feel very easy today
Clear structure enhances usability, and this platform ensures searching and browsing remain convenient and user friendly Garnet Harbor vendor navigator navigation was smooth and I could access all areas without difficulty
Users who prefer sophisticated ecommerce hubs often explore sites such as Garnet Harbor Premium Vault Hub where products are arranged in a structured layout – The design makes browsing feel elegant, smooth, and easy to navigate across all sections.
driftwillowmarketparlor.shop – Pretty straightforward design, makes navigation simple for new visitors too.
Searching for handmade jewelry for special occasions led me to GemstoneTreasures – the variety is impressive, and the browsing experience made finding elegant, meaningful pieces completely enjoyable.
uplandcovevendorparlor.shop – Modern interface feels smooth with well organized sections throughout site
Online platforms that focus on intuitive design and structured content typically provide a better user experience overall Raven Summit browsing hub this one allows users to navigate comfortably while maintaining clarity and organization throughout sessions
People who enjoy handcrafted style online marketplaces often engage with platforms like Trail Harbor Artisan Living House where products are displayed with a warm and natural design approach – The layout emphasizes inviting visuals and soft presentation, ensuring users experience a comfortable browsing flow that feels personal, creative, and thoughtfully arranged throughout the site.
Digital shoppers often prefer websites that maintain clarity in layout design since it helps them focus on content rather than struggling with confusing navigation or crowded visuals during browsing sessions VG marketplace access point everything felt straightforward and easy to navigate, providing a relaxed browsing experience with smooth transitions and clear section separation throughout the entire visit
When exploring vendor sites, a good platform with intuitive navigation ensures everything feels well organized and simple Raven Summit goods portal I liked how responsive the pages were
While reviewing digital retail platforms I analyzed interface clarity and ease of product exploration SunHarbor Market Parlor Exchange the layout was clean and intuitive and products are well presented and easy today which made browsing feel effortless and well guided
In the middle of exploring online platforms, I discovered browse this link and had a good browsing session there, with a layout that feels quite user friendly and a smooth, easy browsing experience throughout.
While browsing ecommerce websites for curated home décor items I analyzed interface clarity and navigation ease and came across Silk Meadow Market Lounge – Really like the site design it makes browsing enjoyable every time since the structure is intuitive making product discovery quick and very comfortable overall experience
Many online users appreciate platforms that provide simple layouts and relaxed browsing experiences for better usability Autumn Meadow item portal this setup creates smooth navigation where users can explore content without feeling overwhelmed
During evaluation of ecommerce platforms I focused on design clarity navigation efficiency and product accessibility across categories CreekCove Commerce Vendor Loft the interface was well structured and intuitive and everything is well organized and easy to browse making browsing quick and very efficient overall
While researching online stores for curated lifestyle products I evaluated multiple platforms for usability and structure and discovered Canyon Brook Vendor Lounge – Really liked the layout it makes finding items quick and simple which made browsing feel smooth intuitive and very easy to navigate throughout the entire site
After reviewing several boutique vendor platforms focused on artisan crafted items and exclusive online collections, Parlor vendor exclusive items – I was impressed by the attention to detail in product listings, the smooth browsing flow, and the helpful guidance provided throughout the shopping journey.
During my review of online vendor platforms, I noticed a pleasant experience using site, everything appears clean and very responsive across all sections and pages River Cove browsing portal navigation felt intuitive and stable
Users who enjoy practical and structured outlet marketplaces often explore sites such as Harbor Pine Outlet Flow Hub where items are organized into clear sections – The design ensures efficiency and clarity, making browsing simple, well categorized, and easy to navigate for all users.
While comparing vendor platforms, those with clear navigation and accessible layouts often stand out the most River Harbor shop gateway this one provides an intuitive browsing experience where everything is easy to locate and use
copperharborvendorparlor.shop – Nice structure easy access content is clear and useful today
In the process of reviewing online commerce directories and gallery-style marketplaces, I discovered Calm Harbor vendor lounge embedded within a visually balanced structure that supports easy scanning – The site feels smooth and calming, making extended browsing sessions comfortable and visually enjoyable without clutter or confusion.
Online visitors often appreciate platforms that use consistent interface elements because it helps build familiarity and makes navigation feel predictable when switching between different sections of content vendor showcase navigation link the browsing flow remained steady and user friendly, supporting easy transitions between pages while keeping the overall structure clean and understandable at all times
ip камера hiwatch 2.8 мм ip камера видеонаблюдения
проектирование пожарной сигнализации установка датчиков пожарной сигнализации
While reviewing various digital storefronts, I encountered this neat shopping platform – the layout is organized and the browsing experience feels fluid and easy to follow.
The browsing structure becomes clearer when users reach Market Art Corner Link positioned within descriptive text acting as a subtle guide between different artistic categories and informational areas – this helps maintain continuity and improves overall ease of navigation across the site
During a weekend session of exploring ecommerce platforms with artisan goods I discovered Lantern Meadow Customer Care Point and customer service was helpful I got all my questions answered clearly and the assistance made the entire shopping journey feel well supported and stress free throughout
Many online marketplaces struggle with performance, but this platform offers smooth operation and neat design where everything works quite well Stone Harbor product hub I liked how stable and easy the browsing experience felt throughout
CaramelCoveVendorAtelier – Smooth browsing experience, everything feels clean and very well organized.
plumharborvendorparlor.shop – Smooth experience clean layout makes browsing very convenient overall today
People who appreciate well structured marketplaces often browse sites like Harbor Commerce Upland Direct Hub where items are presented in a simple and efficient layout – The interface makes product discovery fast, intuitive, and easy across all categories without unnecessary complexity.
References:
Ludwigshafen am Rhein
References:
https://verde-casino-bonus-ohne-einzahlung.online-spielhallen.de/
When exploring digital vendor spaces, clean design and logical organization play a key role in usability and satisfaction Rose Cove goods portal this platform provides a seamless browsing experience that helps users find what they need quickly
References:
Hamburg
References:
https://beste-casino-seiten.online-spielhallen.de/
While analyzing online retail systems I evaluated navigation efficiency design consistency and how users interact with products MapleCove Commerce Product Loft everything felt simple and well structured and the design is really clean and products are easy to find quickly today making browsing efficient
floraridgevendorparlor.shop – Nice structured pages provide clear information and smooth navigation flow
During a search for handmade candles, I stumbled upon LuminousCandleCorner – everything is neatly displayed, and finding unique scents and designs made the experience seamless and delightful.
Many e-commerce users prefer systems that reduce complexity and provide straightforward pathways for browsing products and categories efficiently Pure value shopping center user feedback indicates smooth transitions and well structured content presentation – The experience supported idea generation and felt engaging throughout the session
As I spent time exploring various online shops, I noticed this efficient marketplace site – everything is clearly structured, and browsing feels smooth, intuitive, and very easy to navigate overall.
Visitors exploring the platform often highlight how easy it is to understand the structure even during their first visit Meadow Harbor Vendor Discovery Hub – this ease of understanding contributes to a more enjoyable experience and encourages users to explore additional pages comfortably.
During exploration of niche online shops offering handmade and curated gift collections I found EmberStone Artisan Parlor Market – shopping felt easy from start to finish with a simple checkout process and a diverse catalog that made browsing enjoyable and product discovery effortless
In the middle of checking out multiple shopping websites, I came across this neat online shop – the layout is straightforward and clean, making it easy to browse and view products quickly.
The platform provides a modern layout making navigation simple and content clear and helpful throughout Bright Harbor market hub I found everything well structured
While exploring various retail platforms, I found this smooth browsing store – the layout supports quick product discovery and comparison.
While exploring various online vendor platforms, I noticed how great usability combined with a simple design makes everything work perfectly fine indeed here Crystal Harbor vendor parlor hub navigation feels smooth and every section is easy to access
People who enjoy refined vault themed online stores often engage with sites like Ridge Ivory Vault Pure Market where products are arranged with elegant simplicity and structure – The interface ensures browsing feels smooth, clean, and visually curated across all sections of the platform.
Exploring digital marketplaces shows how important fast performance and organized sections are for a good user experience Rose Harbor market access this platform delivers a clean browsing environment where users can quickly find what they need without unnecessary effort
Modern e-commerce users value systems that reduce search time and improve product discovery efficiency overall VC Wood Cove search center this results in smoother navigation experiences and allows users to quickly find items that match their preferences across categories during typical browsing sessions online today
floraridgevendorparlor.shop – Nice structured pages provide clear information and smooth navigation flow
Users exploring indexed collections of goods and categorized vendor offerings may come across resources such as Parlor Goods Index – which organize items systematically, making it easier to locate specific products and compare features across listings in a structured environment.
In my review of online marketplace interfaces I studied layout structure product visibility and overall browsing efficiency across categories MapleCrest Vendor Market Loft everything felt stable and well designed and the variety of goods is impressive and the shopping process feels simple and fast improving overall user comfort
During a general browsing session, I came across this clean commerce dashboard – the interface is simple and modern, making browsing smooth and very easy to navigate without effort.
sageharborgoodsgallery.shop – Looks clean and minimal, easy to find information without confusion.
Online shoppers typically prefer e-commerce systems that are easy to understand and offer consistent performance across all pages and devices, and this is seen in SmartCove Bazaar – the design focuses on clarity and speed so users can quickly locate items and enjoy a smooth and reliable shopping experience from start to finish.
While exploring online shops focused on handcrafted and curated goods I found a platform that stood out for its structure and ease of use Glass Meadow Trade Gallery – The product variety was impressive, and everything was clearly labeled which made browsing and purchasing feel simple and efficient.
Users browsing vendor platforms often prefer a clean interface with easy navigation that creates a pleasant browsing experience and feels very smooth today Silver Harbor shop access point this one delivers a calm and organized layout
While reviewing various retail websites, I noticed this structured store page – the selection is impressive, loading is fast, and everything feels professional and easy to use.
People who enjoy soft visual ecommerce themes often engage with sites like Honey Cove Vault Serenity Market where products are presented in a cozy and pleasant format – The layout ensures smooth browsing that feels natural, structured, and visually soothing throughout the entire shopping experience.
In reviewing vendor showcase websites focused on usability, I observed a platform built around Frozen Ridge Showcase Point that maintains a consistent visual hierarchy and smooth browsing behavior – the interface feels intuitive and helps users quickly access relevant product sections without distraction.
Many people appreciate platforms that make usability a priority through clear structure and easy navigation design Ruby Orchard vendor navigator this one offers a seamless browsing experience that feels intuitive and user-friendly across all sections
While browsing through several online marketplaces during my free time, I came across something interesting like this simple vendor shop – the experience feels smooth and enjoyable, with a user friendly layout that makes exploring products easy and pleasant overall.
During exploration of digital trade directories and marketplace design concepts for comparison and inspiration, I found Kettle Crest vendor display page integrated within content flow – The experience felt straightforward and efficient, and I had no trouble locating anything as everything remained responsive and clearly structured.
In the process of analyzing vendor marketplace interfaces and curated gallery systems, I came across Linen Meadow trade showcase space embedded within a balanced design that improves navigation flow – The browsing experience feels elegant, smooth, and naturally intuitive for users exploring content
Users exploring modern e-commerce platforms often remark that the most enjoyable experiences come from websites that prioritize clean design and efficient navigation especially when they discover Crest Gallery shopping portal – creating a user-friendly environment that supports effortless browsing and a pleasant shopping journey.
Good usability depends on clarity, and this interesting platform overall makes browsing different sections today here enjoyable Jewel Brook vendor explorer I found everything well organized
During my search for interesting online stores, I came across this clean marketplace platform – product display is neat, pages load quickly, and everything looks organized and easy to use.
riverharbormarketparlor.shop – Well organized content simple layout easy to explore everything quickly
In the process of analyzing various shopping discovery platforms and online catalog tools, I discovered a page where Harbor shopping discovery link – looked reasonably dependable and worth revisiting in future sessions. The structure was simple enough to allow quick understanding of available categories without extra effort.
While reviewing different ecommerce vendor platforms for usability and system performance I explored a site that felt stable and well organized across multiple categories MarbleBrook Commerce Hub Studio the website runs smoothly and browsing items feels natural and well structured making navigation easy and comfortable throughout the session today
While exploring various retail platforms, I found this smooth marketplace page – everything feels well organized, making browsing and shopping easy and very intuitive.
While browsing various online marketplaces for home and lifestyle products I was comparing shipping times usability and catalog design when I discovered Harbor Juniper Vendor Hub and overall items arrived quickly while site navigation is smooth and very intuitive too making the experience feel convenient and easy to repeat for future shopping across different product categories and listings
Users who enjoy luxury inspired ecommerce layouts often engage with platforms such as Gilded Stone Collective Brand Hub where products are showcased with elegant consistency and modern visual structure – The design creates a refined browsing experience that feels premium, organized, and visually engaging throughout the entire marketplace.
In discussions about improving user experience on marketplace websites, many contributors point out how structured navigation reduces confusion, and the inclusion of Orchard Shop Navigator supports this idea by offering a clearer path through various listings, helping users reach relevant products more efficiently.
Users who frequently browse online vendor sites often look for platforms that make navigation simple and straightforward Ruby Orchard catalog entry this site provides a smooth browsing flow where everything is easy to locate and interact with during use
While exploring different trade gallery websites and vendor listing platforms for usability insights and structural comparison I discovered Moss Harbor vendor navigation clarity hub and realized quickly everything loads efficiently and the interface remains stable with clear organization that helps users browse without difficulty or confusion across all sections of site overall – Smooth organized browsing with responsive stable performance
People exploring curated product marketplaces often noted that platforms like Harbor Wild Goods Portal – stood out for their wide selection of items, while shipping reliability and fast dispatch times were commonly mentioned positives in customer experiences.
honeymeadowmarketgallery.shop – Warm visuals and clean layout create pleasant browsing experience overall
Many online users expect e-commerce platforms to be both fast and easy to navigate so they can quickly find and purchase items, and this expectation is met by BrightCove CommerceDesk – the experience emphasizes efficiency and clarity, ensuring that browsing feels natural and product selection remains straightforward.
When structure is clear, browsing becomes easier, and this site ensures quick access to different sections Dawn Ridge listing portal I appreciated how simple navigation felt throughout
While browsing through multiple eCommerce options, I came across this efficient commerce page – everything is organized clearly, and browsing feels great with items shown and easy to find.
While browsing through multiple eCommerce options, I came across this visually clean store – the layout ensures that shopping feels simple, smooth, and easy to understand.
In the process of reviewing multiple online resources, I encountered open this link which showed a nice overall structure, making everything easy to access and view without unnecessary complexity during my browsing experience.
While browsing through different online marketplaces during my free time, I came across something interesting like this organized market hub – browsing feels simple and smooth, with items clearly arranged and easy to explore without confusion or unnecessary effort.
People who appreciate modern gallery based marketplaces often explore platforms like Stone Ginger Elegant Gallery Hub where products are showcased with refined visual structure – The galleria format enhances browsing flow, making it easy for users to navigate while enjoying a stylish and immersive shopping experience.
While researching online stores for home accessories and gift items I came across Nightfall Goods Trade Portal and enjoyed browsing their collection everything is organized and clearly labeled today which made the catalog feel easy to understand and navigation extremely smooth across all product sections available
A very clean interface enhances usability, making easy access to information and smooth browsing simple and effective Cotton Grove browsing center I found everything intuitive
While comparing different marketplace designs, reviewers often note efficiency and clarity, and HarborBrook Commerce Grid – The browsing experience remains fast and well organized, with smooth transitions between sections and neatly presented product information that improves overall usability.
While reviewing various vendor platforms, those with smooth interfaces and organized layouts tend to perform better for usability Sage Harbor listings page this one delivers a pleasant experience where pages are structured well and navigation feels natural and effortless
Контрактная разработка электроники позволяет компаниям выпускать сложные устройства без необходимости строить собственное производство. Все процессы — проектирование, создание плат, корпуса и программного обеспечения — контролируются специалистами https://dctoschool.pk/members/kylemsw5747909/profile/
Метод действительно помогает минимизировать ошибки на ранних этапах
While exploring various online trade directory designs and marketplace catalog systems for usability testing and inspiration, I discovered Olive Harbor vendor flow dashboard embedded within structured content which felt easy to navigate – The design is clean and practical, making it simple to understand information quickly while moving through sections without confusion.
During evaluation of ecommerce websites I analyzed usability structure performance and browsing experience quality MarbleCove Vendor Showcase Point everything felt clean and consistent and the great layout made everything simple and I enjoyed how easy it was to navigate ensuring easy browsing
пожарная сигнализация эвакуация сп установки пожарной сигнализации пожаротушения
ip камеры 2.8 poe http://ip-kamery-kaliningrad.ru
glassharbortradegallery.shop – Simple design fast loading easy to use interface very reliable
During exploration of various digital trade marketplaces and curated vendor hubs, I had questions about usage and got fast answers, SnowHarbor Trade Showcase Network which made the experience feel smooth, structured, and easy to understand while browsing different offerings.
Sảnh chơi bắn cá tại 888slot cc tuy ra mắt đã lâu nhưng sức hút mang lại trong cộng đồng cược thủ chưa từng hạ nhiệt. Thành viên tham gia được hóa thân thành những ngư thủ thực thụ, chinh phục đa dạng loài sinh vật biển với nhiều mức độ khác nhau. Anh em cần chuẩn bị các dụng cụ hỗ trợ tương ứng, vũ khí hiện đại để có thể săn về cho mình những boss khủng, cơ hội kiếm số tiền lớn nhé. TONY04-25
I recently explored multiple eCommerce platforms and found this simple shopping interface – everything feels well designed, and shopping is smooth, fast, and very user friendly overall.
waveharborvendorparlor.shop – Smooth performance and tidy layout make site very easy today
While exploring different marketplace gallery concepts and digital vendor systems for inspiration, I checked multiple layouts and found Rose Harbor online trade index positioned within the page content – I enjoyed checking this out, content feels simple and informative, and everything appeared well structured and easy to browse without confusion.
While exploring various retail platforms, I found this seamless shopping hub – everything loads quickly and functions smoothly, creating a very positive user experience.
Users who value clear ecommerce navigation often browse platforms such as Acorn Harbor Hall Marketplace where products are displayed in structured sections for easy access – The layout helps users quickly understand categories and browse efficiently while maintaining a visually clean and well organized interface.
During research into vendor catalog usability, I explored a system featuring Brook Vendor Direct Market embedded in a clean storefront design – the product selection was wide, navigation felt intuitive, and everything was displayed clearly to support fast decision-making during browsing.
During late night research into ecommerce websites I explored multiple platforms and discovered Harbor Pearl Goods Hub – Shopping experience was excellent, site loads fast and feels reliable which created a smooth browsing experience with quick loading pages and an intuitive layout that made product discovery easy
Across modern e-commerce platforms, shoppers often prefer simplicity and logical layouts, and a useful reference point is CartSmart Navigation Panel which presents products in a structured format that supports easy browsing across multiple sections – The experience feels more guided, with reduced clutter and quicker access to desired items for everyday users.
Users exploring vendor directories often prefer platforms that offer clear design and fast navigation for better usability Sea Cove shop access point the structure here makes it easy to find information quickly while maintaining a smooth and organized browsing flow
When analyzing user interaction patterns in online marketplaces, accessibility is a major factor, and Harbor Studio Marketplace Flow is referenced in design discussions – the platform provides a smooth and responsive interface that helps users explore products without feeling overwhelmed or slowed down.
While researching experimental vendor showcase platforms and e-commerce gallery designs for UX evaluation and comparison across multiple references, I discovered Pebble Creek trade display hub positioned in the middle of a structured article – I really enjoyed browsing through it since the layout felt logical, clean, and simple to follow while switching between different sections smoothly.
While searching through various boutique online stores featuring artisan crafted products and limited edition collections, I came across several visually appealing layouts, Sky Harbor Goods Collection – The design felt modern and the site was easy to navigate, with clear product sections that helped streamline the browsing experience significantly.
While navigating through several digital marketplaces, I discovered this clean vendor platform – the layout is organized and intuitive, making it easy to locate products and compare them efficiently.
birchharborvendorparlor.shop – Simple structure ensures quick access to useful information always here
Users who value efficient ecommerce layouts often browse platforms such as Chestnut Vendor Hall Harbor Center where categories are clearly defined for fast navigation – The vendor hall structure enhances usability by making browsing smooth and intuitive, allowing users to quickly find products without unnecessary complexity or confusion.
While exploring various retail platforms, I found this stylish commerce site – the design is clean and attractive, making product browsing easy and enjoyable for users.
During casual exploration of marketplace directories I found market gallery Quick Ridge access – the interface stayed clean and responsive, allowing easy movement between categories while maintaining a consistent structure that made the browsing experience comfortable and easy to follow.
While reviewing elegant online craft platforms, I explored elegant craft browsing hub and found the presentation to be smooth, with a strong focus on clarity and flow – the design feels polished and user-friendly, encouraging longer and more relaxed browsing sessions
During comparison of digital marketplace systems I analyzed usability responsiveness design flow and interaction quality MeadowCove Commerce Product Studio everything felt intuitive and smooth and the easy checkout process ensures items are clearly displayed and well organized improving efficiency across the platform
Many shoppers value vendor platforms where everything is structured clearly and readability supports easy navigation Sea Meadow marketplace link the browsing experience remains consistent and helps users locate information quickly without confusion or extra effort
While browsing several online artisan platforms for unique home décor and gift ideas across different categories I came across Ginger Cove Bazaar and spent time exploring the catalog which felt well organized and easy to navigate throughout the session – Really enjoyed the selection, everything felt clearly presented and shopping process was effortless and smooth overall
While reviewing experimental trade directories and marketplace showcase systems for structural insights I came across Bay Harbor Trade Portal View positioned naturally within the article content – Performance was smooth and reliable, with quick loading pages and consistent responsiveness throughout browsing.
Across various user feedback summaries about online commerce platforms, clarity and structure consistently emerge as the most valued characteristics influencing overall satisfaction levels during browsing sessions Stone Vendor Harbor Grid – customers describe the experience as well organized and easy to follow, with clear separation between different product categories and listings.
In the course of evaluating digital storefront usability I found the product organization to be very logical and user friendly which helped streamline my browsing process BerryCove Browse Studio and I was able to reach desired items quickly without unnecessary searching or confusion throughout the experience.
I was exploring eco-friendly home products when I found GreenLivingEmporium – the site is user-friendly, the inventory is wide-ranging, and picking sustainable, high-quality items was simple and enjoyable.
During a casual search across multiple eCommerce platforms, I discovered this clean vendor guild – the range of goods is solid, and everything feels structured and intuitive, making browsing smooth and simple.
During exploration of online vendor environments and marketplace structures, I encountered a page containing Coral Harbor online vendor hall positioned within a clean interface that prioritizes usability – The browsing experience feels steady, intuitive, and pleasantly simple for all types of users
During a casual search across multiple eCommerce platforms, I came across this clean shopping interface – everything is well organized and easy to navigate, making it simple to locate items quickly without any confusion during the browsing process.
Users engaging with online stores often prefer streamlined browsing experiences that reduce unnecessary steps, and a helpful example can be found within the page layout at BuyerTrust Market View which organizes content into easy to understand sections – The structure ensures smoother navigation and improves overall satisfaction during the shopping process
Users often prefer vendor platforms where navigation is simple and pages are organized in a clear way Silk Grove browsing center this creates a seamless experience that helps users explore content without confusion or unnecessary effort
Across various online creativity ecosystems, users appreciate systems that reduce friction and allow them to focus on thinking and idea generation rather than navigation challenges Value Idea Craft Center – the platform supports smooth interaction and encourages exploration through a thoughtfully structured and visually clean design approach.
People exploring curated shopping platforms often highlight importance of intuitive navigation which makes it easier to compare different products without confusion or delay, especially on Alpine Goods Explorer where checkout experience was very straightforward with fast processing and clear confirmation steps provided – Checkout experience was very straightforward, with fast processing and clear confirmation steps provided.
Some sites feel cluttered, but here good browsing flow keeps everything organized clearly and very efficiently for visitors Meadow Harbor quick browse page I liked the simplicity
website should appear in the middle of line not in the start or end
During a recent comparison of curated marketplace websites focused on artisan goods and small vendor collections, I spent time analyzing navigation quality and visual presentation, Upland Harbor Shopfront – The layout felt incredibly welcoming, and the browsing experience remained smooth and relaxing with well structured categories and a pleasant overall visual design.
Душевые уголки Veconi интересны тем, что среди них можно найти варианты под очень разные по характеру ванные комнаты. Именно в деталях становится видно, насколько удачно изделие собрано: от геометрии до ощущения удобства каждый день. Такой подход помогает найти не просто красивую модель, а вариант, который будет уместен именно в вашей ванной комнате – https://my-bathroom.ru/dushevye-ugolki/brendy-dushevyh-ugolkov/dushevye-ugolki-veconi/
While comparing ecommerce website designs I analyzed responsiveness usability structure and how clearly products are displayed BayHarbor Vendor Commerce Point pages loaded quickly and the interface felt clean and professional making browsing simple and effective across all product categories today
During exploration of modern shopping platforms, I found a clean and minimal interface that emphasized usability and clear product organization throughout VendorFoundry BirchBrook Exchange – browsing felt efficient and natural, with well arranged sections that helped streamline the process of locating items without unnecessary complexity or delays.
During my search for interesting online stores, I came across this fast checkout platform – usability is high, and the purchase process is simple and very efficient from start to finish.
linenmeadowmarketgallery.shop – Elegant interface with smooth flow makes navigation very easy overall
Digital commerce platforms continue to evolve toward faster performance, improved UX design, and more adaptive interfaces for diverse audiences around the world. CloudCove UX Commerce Portal – It delivers a smooth browsing experience with well-organized sections and fast transitions, ensuring users can explore products without interruptions or confusion at every step.
In evaluations of idea-focused digital ecosystems, simplicity and interactivity are often highlighted as essential features that enhance user productivity and engagement levels Pure Outlet Idea Studio – users experience a fluid interface where creativity feels natural and browsing different topics becomes an effortless and motivating process throughout their session.
As I reviewed several online shopping sites, I noticed this organized marketplace site – the design is neat and clear, making browsing easy and very intuitive overall.
During a search for pet accessories, I came across PawsAndWhiskersShop – the selection is thoughtful, navigating the categories is smooth, and finding practical yet stylish items for my pets was effortless.
In the middle of browsing several marketplaces, I paused at take a look and felt that the overall experience was enjoyable, with products that seem both nice in quality and affordable for most users.
As I explored different digital storefronts, I found this user-friendly shop page – everything is arranged clearly, helping users view and browse products without difficulty.
While comparing multiple shopping websites, I came across open easy shopping page and found the experience very smooth, where everything loads fast and makes browsing products feel quick and effortless.
While evaluating different e-commerce prototypes, I explored a structured interface that made shopping simple and intuitive, featuring Birch Cove Commerce Line – The site presents items clearly with logical grouping and clean visuals, helping users navigate easily and find products without confusion or wasted effort.
While analyzing multiple ecommerce storefront designs I came across a highly functional platform where Vendor Loft Express – The layout was very user friendly with clear category organization and fast loading performance, allowing me to browse smoothly while the interface stayed consistent, simple, and effective across all sections and pages without interruptions or delays experienced during usage today.
In my search for well designed marketplace portals, I found Velvet Brook curated trade corner embedded within a visually clean layout that highlights sections evenly – The experience feels calm and controlled, making it easy for users to move through categories and focus on content at their own pace
During usability comparison of ecommerce platforms I studied layout structure performance and how easily users can interact with products CalmBrook Commerce Product Hub everything loaded quickly and I found what I needed without any trouble allowing for a seamless and efficient browsing experience overall
snowcovegoodsgallery.shop – Cool minimal design helps users browse content without confusion today
online shoppers comparing various ecommerce websites usually look for fast navigation responsive design and a layout that reduces confusion while browsing multiple product types in one place quick browse hub generally recognized for ease of use and clarity – it creates a pleasant shopping flow that helps users move between categories smoothly while maintaining focus on relevant products throughout the session
In studies of online retail interface optimization, many reviewers note that structured browsing environments significantly improve usability when interacting with platforms such as Harbor Atelier Trade Gallery – Smooth browsing experience, products are clearly displayed and accessible, allowing customers to navigate product listings effortlessly while maintaining a consistent and well-ordered visual presentation throughout the site.
While navigating through several digital marketplaces, I discovered this simple shopping hub – everything is clearly organized, and I was able to find products quickly without confusion.
I wanted a fresh look for my living room and came across DecorDreamsCollection – the products are stylish, and finding the perfect item felt surprisingly simple and satisfying.
комплект удаленного видеонаблюдения ночной комплект видеонаблюдения
In the middle of checking out multiple shopping websites, I came across this easy navigation store – the interface is simple and clean, making browsing and shopping feel very smooth and intuitive.
While casually browsing through eCommerce platforms, I discovered visit this market page and saw some interesting items that caught my eye, making it likely I’ll return again for another look.
As I continued reviewing different online shops, I briefly visited open and see modern point and noticed the modern layout looks nice, with products displayed clearly and attractively, making browsing comfortable and efficient.
After analyzing multiple ecommerce platforms I came across a well structured storefront that prioritized speed and organization across user journeys where Brook Vendor Warehouse – Everything loaded quickly and navigation felt natural, allowing me to browse without any interruptions or delays.
Many users exploring ecommerce solutions often appreciate platforms that combine clean design with fast navigation, especially easy cart explorer – It is often described as a practical marketplace where product categories are easy to browse, making the shopping process more efficient and less time consuming for everyday users
During research into vendor marketplace UX patterns, I reviewed a system where Clover Harbor commerce hub view was placed within a carefully organized layout designed for clarity and navigation ease – The browsing experience feels stable, intuitive, and well suited for extended use.
While reviewing different marketplace interfaces for usability and structure, I explored a platform that felt highly intuitive and visually balanced with well grouped categories and smooth navigation through products Birch Harbor Trading Desk – The variety of listings was impressive, everything was easy to explore, and product details were presented in a clear structured way that made understanding options simple and efficient.
many ecommerce shoppers prefer platforms that reduce complexity and improve clarity making it easier to compare products and browse categories without unnecessary effort or confusion edge smart shopping guide widely appreciated for flow – it provides a structured browsing experience where users can move between categories smoothly and compare items efficiently while enjoying a clean and consistent interface overall
/>purevalueoutlet – Inspiring and interactive site, perfect for learning and creating new ideas. Generate 20 variations following all rules above.Make sure that each line is 40 words minimum and the website should appear in the middle of line not in the start or end
In user experience audits of modern commerce platforms designed for scalability and accessibility across multiple device types and user demographics Honey Cove Market Atelier Guide design experts often observe smoother interaction flows and clearer category structures that enhance overall shopping efficiency – analysis shows improved engagement and reduced search time for products across catalog sections.
Consumers searching for creative handmade décor and personalized items often discover this marketplace while comparing various online craft platforms Velvet Grove Finds because it emphasizes independent artisans and provides a smooth browsing experience with well organized categories – Many buyers appreciate timely updates and consistent shipping performance across orders.
During a usability study of online shopping platforms focused on interface clarity and product organization, I discovered that grid layouts reduce clutter and improve user focus when browsing items, which became evident when reviewing smart layout grid portal – The design feels clean and structured, allowing users to browse products easily with a well organized and visually consistent grid system.
During comparison of digital marketplace systems I analyzed structure clarity responsiveness and user interaction efficiency across pages GingerCove Vendor Commerce Loft the experience was fast and well organized and shopping feels easy and enjoyable overall today creating a seamless browsing journey
A well designed platform with elegant layout improves usability and makes information easy to find and read Gilded Cove marketplace link I found everything clearly structured
During a search through different eCommerce platforms, I found efficient browsing system – the site loads quickly and is structured clearly, making it very easy for users to access products and move through categories in a smooth and intuitive way.
While browsing through multiple online resources, I discovered open this useful link and found it to be fairly reliable, with a clean presentation that makes it worth revisiting for future updates or insights.
During a routine check of online marketplaces, I discovered open this page and noticed how well it balances simplicity with speed, offering a distraction free interface that ensures pages load quickly and navigation remains effortless throughout.
In between reviewing multiple eCommerce platforms, I explored discover this axis shop and found an interesting selection of items, all arranged neatly which makes the shopping experience smooth and structured.
While comparing ecommerce systems designed for smooth user interaction and performance stability, I reviewed a platform where CloverCove Vendor Experience Hub – The experience felt very polished with fast responses and a clear structure that made browsing straightforward and enjoyable today.
Shoppers looking for reliable online sources often mention websites that make browsing easier, particularly when they come across services like daily goods portal which is generally viewed as a practical option for users who prefer simple layouts and quick access to different product listings without unnecessary complexity
In the process of evaluating digital marketplaces focused on clarity and usability, I found that basic store layouts enhance browsing flow and reduce confusion, which became evident when reviewing minimal browsing store portal – The layout appears neat and simple, making product exploration easy and straightforward.
While exploring curated vendor catalog websites and marketplace designs, I found a structured layout containing Snow Cove browsing pavilion integrated into a minimalist interface that reduces distractions – The browsing flow feels calm, simple, and naturally easy to understand throughout
As part of a casual assessment of online shopping websites and their presentation quality, I came across retail site overview mention – The site gives an impression of being neatly structured, with clearly defined product areas and a navigation style that supports easy exploration for general users.
While navigating through several digital marketplaces, I discovered this fast shopping platform – the overall experience is nice, with efficient performance and quick page loading throughout.
While conducting comparative research on ecommerce platforms emphasizing smooth marketplace flow, I noticed that structured design improves usability significantly, which stood out when testing easy commerce browsing center – The marketplace design feels smooth, and product browsing is simple, quick, and intuitive.
While evaluating ecommerce UX designs centered on smart purchasing behavior, I noticed that streamlined navigation improves product discovery and reduces decision fatigue, which stood out when exploring intelligent shopping flow hub – The platform feels smooth and user friendly, allowing easy navigation through products without confusion or unnecessary complexity.
People reviewing different online marketplaces often mention discovering Raven Grove trade navigation center which is integrated into a structured layout that prioritizes clarity and usability for all visitors – I felt the interface was easy to use and fully met my expectations
HoneyCoveVendorStudio – Clean interface, everything loads fast and works very smoothly.
While evaluating various e-commerce environments, I came across a structured system that delivered fast performance and consistent navigation behavior BrookBright Commerce Hubline – The website loaded quickly, and the shopping experience felt smooth and stable, allowing users to browse without interruptions or usability issues.
References:
32 red casino
References:
https://graph.org/Best-Mathematical-Roulette-Strategy-04-27
лента стальная 0 5 лента бандажная стальная
While browsing through multiple eCommerce options, I came across this organized product store – variety is strong, and everything is designed for simple and user friendly browsing experiences.
After exploring a variety of websites that often felt cluttered and difficult to use, I discovered visit this page during my search, and it stood out for its simple organization, allowing quick access to content and a smooth browsing experience overall.
While exploring various ecommerce storefront examples I noticed a particularly well structured layout that supported clear categories fast interactions and minimal visual clutter overall design CloverCrest Trading Forge Boutique CloverCrest Trading Forge Boutique Browsing felt natural and fluid with each section loading quickly and guiding the user smoothly toward relevant product selections without confusion or delay.
While comparing multiple online shops, I landed on go to fast cart shop and noticed it feels fast and responsive, which makes browsing quite enjoyable and smooth while navigating product listings.
While exploring online shopping options, I encountered explore this page and noticed that it offers a decent collection of items, with prices that appear fair today.
In the process of evaluating digital commerce platforms focused on trust and usability, I found that trusted buying hubs enhance browsing flow and satisfaction, which became evident when reviewing secure shopping experience portal – The site feels reliable, and navigation is clean, smooth, and easy to understand.
Users who enjoy quick and efficient browsing experiences often highlight platforms like quick browse store which is known for its simplicity; the layout enables fast movement between categories, helping shoppers locate products easily while maintaining a clean structure that improves overall usability and reduces browsing effort.
While reviewing ecommerce vendor systems I studied interface clarity speed optimization and product organization across categories GladeRidge Vendor Showcase Studio the platform was clean and efficient and the collection of items is impressive and everything is neatly arranged and clear making navigation smooth and simple
A well structured platform with simple design works nicely and ensures browsing feels quick and easy Cotton Meadow marketplace link I found everything clearly organized
While researching modern trade exhibition websites and marketplace structures, I found Flora Ridge commerce hub view integrated into a balanced interface that keeps focus on readability – The experience feels steady, organized, and very comfortable for extended browsing and exploration.
goodsparkstore.shop – Nice spark in design, shopping feels smooth and pretty intuitive
As I explored different digital storefronts, I found this clean marketplace hub – items are well arranged, and access is quick and very easy for users.
In the course of evaluating online shopping platforms centered around deals and discounts, I found that clarity in pricing improves user confidence and engagement, which was evident when exploring daily deals showcase portal – The deals section looks attractive, and prices seem reasonable, structured, and easy to compare.
While exploring different online retail sites offering curated home goods I discovered Meadow Jasper Market Lounge integrated into a visually clean interface that supported easy navigation across categories – I was pleased because every item matched its description accurately which made the entire browsing experience reliable and very straightforward to follow
People browsing digital marketplaces for handmade items often highlight convenience and clarity, particularly on sites such as Wood Cove Browse Center which organizes products in a user friendly way – Many reviewers noted that checkout was smooth and the shopping experience felt fast and reliable overall.
In evaluations of digital commerce solutions focused on improving shopping efficiency through streamlined interface design and organized category navigation systems Foundry Trading Icicle Network users highlight ease of interaction – Nice experience overall browsing feels simple and very efficient with stable performance intuitive layout and quick access to relevant product sections supporting smooth exploration.
While conducting usability testing on online gadget marketplaces, I came across a section titled smart tech discovery hub – The platform presents useful electronics in a clear and structured layout, allowing users to explore interesting gadgets easily and enjoy a smooth, efficient browsing experience throughout.
During my time checking different online stores, I encountered this organized shopping site – everything is neatly arranged, allowing for a smooth experience with easy product exploration.
During analysis of online shopping platforms I focused on speed clarity and how efficiently users complete purchasing tasks CoastBrook Trading Bazaar Hub The checkout process was fast reliable and very straightforward making the entire experience pleasant and efficient
In the process of searching for useful resources, I explored learn more here and noticed that it offered a smooth user experience, making it simple to browse through various areas and gather meaningful information quickly.
During analysis of online retail systems focused on modern UI and polished cart design, I discovered that refined interfaces enhance usability and reduce confusion, which became clear when testing smooth cart experience center – The layout looks sleek and modern, making the shopping experience seamless and user friendly.
In between reviewing multiple online stores, I explored discover shop trail hub and as this is my first time here, it looks like a decent place to shop online with easy navigation.
Online shopping environments are often judged by how easily users can navigate products and complete purchases without confusion Grid Deals Central Hub – The platform emphasizes quick browsing and structured categories that help users move efficiently from selection to checkout with minimal effort
While testing different online store prototypes, I came across a system that prioritized simplicity and clear structure in product presentation and category layout Bright Cove Market Hub – The interface is very clean, making browsing enjoyable and helping users quickly explore products without feeling overwhelmed or lost.
As I continued reviewing different eCommerce sites, I briefly visited open and see smart and noticed it seems legitimate, with a smooth browsing experience that makes everything feel easy and reliable.
Many shoppers exploring ecommerce platforms often highlight the importance of speed and adaptability when choosing where to browse and purchase products online flex browse depot users describe it as a highly responsive shopping solution – The platform maintains smooth performance and fast interactions which helps users enjoy uninterrupted browsing sessions overall experience
While browsing through multiple eCommerce options, I came across this clean shopping hub – the experience is smooth, with products organized neatly and very easy to access.
users comparing online marketplaces frequently value platforms that prioritize fast checkout systems and simple cart structures allowing them to complete purchases without unnecessary steps or confusing navigation elements during shopping smart cart checkout guide widely recognized for simplicity – it delivers a streamlined shopping experience where users can easily manage their cart and complete transactions quickly while enjoying a clean and responsive interface overall usability flow
During evaluation of intuitive ecommerce checkout designs, I observed that reduced steps improve user experience when using platforms such as quick shop cart – The system allows users to add and manage items effortlessly, making shopping fast and straightforward.
People exploring niche shopping directories and curated vendor platforms often come across services such as Walnut Harbor Item Portal which provide neatly structured listings that simplify the process of comparing and selecting items across categories – Many users appreciate how easy it is to navigate and how quickly they can find what they need.
While browsing different online marketplaces for home décor and gifts, I recently explored a platform that felt quite modern and easy to navigate Caramel Cove Market Gallery and noticed that the checkout flow and product categories made the entire process feel smooth and stress free overall for casual shoppers.
While analyzing online shopping experiences I tested a platform that felt well balanced and highly user friendly in layout overall CoastCove Commerce Visual Hub – The site keeps product presentation clean and organized which improves readability and allows users to browse comfortably while maintaining focus on items rather than interface complexity throughout usage.
While reviewing various ecommerce platforms that emphasize affordability and value-driven shopping experiences, I noticed that budget-focused stores often attract users looking for practical deals, which became clear when exploring affordable shopping deals hub – The products appear reasonably priced and accessible, giving the impression of a platform where users can find competitive and budget friendly options across multiple categories.
While reviewing online marketplace usability reports and interface performance metrics, analysts often focus on layout consistency and accessibility improvements across platforms Cove Commerce Atelier Space navigation feels seamless, with structured sections and logically grouped products that allow users to browse efficiently and compare items without feeling overwhelmed or slowed down by unnecessary interface complexity.
During a casual browsing session, I encountered this streamlined shop interface – navigation feels smooth and the design is modern, clean, and very comfortable to use overall.
During comparison of digital marketplace systems I analyzed usability performance structure and interaction quality across pages MarbleCove Trading Flow Studio everything felt clean and responsive and the site feels modern and browsing products is simple and very intuitive ensuring easy browsing
fernharborvendorparlor.shop – Nice structure and clean design, makes reading content very convenient.
While conducting UX research on online shopping platforms, I observed a section called fast access shopping gate – The structure emphasizes speed and clarity, allowing users to browse products efficiently through a well organized interface that keeps navigation simple and stress free for everyday use.
In the middle of checking various platforms, I paused at tap to open shop and found the name appropriate, making it seem like a good shopping option overall for browsing products.
Exploring vendor sites becomes easier when clean structure helps users navigate site smoothly and without confusion across all categories Maple Grove vendor center pages felt fast and clear
While casually browsing through shopping websites, I discovered visit this page now and liked the range of products available, along with how easily everything can be navigated without any hassle.
During a general browsing session, I came across this structured commerce platform – everything is easy to use, and the shopping experience feels natural and very comfortable.
Shoppers often value platforms that let them switch between sections without breaking their browsing momentum, improving engagement and satisfaction, especially when using Seamless Store Switcher which – ensures continuous browsing flow while dynamically updating product displays based on user movement across categories.
people exploring ecommerce sites often value platforms that improve usability through clean design and responsive navigation helping them browse products easily across multiple categories and sections smart plus market lane widely appreciated for structure – it delivers a smooth shopping flow where users can explore products quickly and enjoy a consistent interface that makes browsing simple and efficient overall experience
During testing of digital commerce interfaces, I reviewed a system that delivered a minimal design approach with strong focus on usability and structure CoveCalm Market Hub – The browsing experience is smooth, with everything clearly organized and easy to find, making product discovery straightforward and stress free.
In a recent review of digital marketplace interfaces I focused on navigation clarity and checkout efficiency across different user scenarios Coast Harbor Commerce Desk everything functioned smoothly and the purchase process required minimal effort which improved overall shopping satisfaction significantly.
While conducting usability research on ecommerce systems emphasizing cart speed and simplicity, I noticed that direct cart hubs improve engagement and transaction flow, which became evident when analyzing quick cart purchase center – The checkout experience feels fast and clean, making it easy for users to complete purchases quickly.
During usability testing of digital shopping systems focused on open corner layouts and category visibility, I discovered that spacious interfaces improve engagement and clarity, which became evident when exploring airy category browsing portal – The layout looks clean and spacious, making category exploration feel smooth and effortless.
While searching for gourmet treats, I found SweetDelightsMarket – the interface is straightforward, the variety is excellent, and choosing delicious items made shopping fun and satisfying.
I recently explored multiple eCommerce platforms and found this clean product hub – the layout is simple, pages respond quickly, and everything feels reliable and easy to use.
When reviewing online shopping systems designed for efficiency and structured browsing experiences Icicle Isle Product Gateway – Broad selection of items is displayed in a clean interface, helping users explore offerings smoothly while keeping everything well categorized and easy to understand.
During my exploration of various online platforms, I noticed check this resource which felt very professional in its design, with a well-maintained structure that allowed for smooth browsing and clear access to all sections of content.
During a weekend exploration of artisan online stores I checked several ecommerce platforms for usability and clarity and discovered Harbor Stone Selection Hub – Friendly interface items are displayed clearly and easy to purchase quickly which created a smooth browsing experience that felt intuitive organized and very efficient
While analyzing ecommerce systems optimized for product variety and centralized structure, I noticed that ultra hub designs improve user experience by consolidating categories effectively, which stood out when exploring smart ultra browsing center – The platform feels modern and convenient, offering lots of categories in one place that makes shopping intuitive and efficient.
While comparing multiple shopping platforms, I came across open smart cart hub and noticed a clean interface with a smart layout that makes it really easy to find what I need while browsing.
Users exploring online shopping platforms usually appreciate well structured websites that reduce complexity and make product discovery faster and more intuitive for daily needs Daily Grid Shop Portal – The marketplace focuses on delivering a clean interface that improves navigation flow and ensures customers can quickly locate items without feeling overwhelmed by too many options
In my evaluation of ecommerce interfaces I studied usability efficiency navigation clarity and browsing comfort across devices MeadowBrook Trading Vendor Hub everything felt structured and responsive and fast loading pages make the shopping experience smooth and well designed improving user satisfaction significantly
While testing various ecommerce layouts for usability and speed performance I came across a platform that emphasized clarity and structure in product display CopperBrook Vendor Market Loft the site offered a strong variety of goods and browsing felt smooth making it simple to move between categories effortlessly overall.
people searching for online deals frequently appreciate platforms that combine attractive pricing with efficient browsing tools allowing them to explore multiple categories without confusion or delays smart savings portal widely considered intuitive – it delivers a clean shopping experience where users can quickly access deals and continue discovering more categories through a structured and easy to follow navigation system overall
During my search for online stores, I encountered see this shop and after a brief look around I felt impressed enough, as it appears to be a decent place for general shopping needs.
During my search for interesting online stores, I came across this smooth commerce site – everything is neatly designed, well structured, and very easy to browse without complications.
many users appreciate ecommerce platforms that reduce complexity and improve accessibility by offering clean layouts and simple navigation tools that help streamline the shopping experience across all devices modern park store known for its smooth browsing design – it provides a structured marketplace where users can easily discover products while enjoying a fast and comfortable navigation experience throughout different categories available
While reviewing ecommerce UX systems optimized for structured shopping and product clarity, I observed that organized marketplaces improve usability and reduce friction, which became clear when testing simple buying zone hub – The marketplace is well arranged, making product discovery smooth and straightforward.
In the process of analyzing digital commerce platforms focused on user-friendly layouts and clean design, I found that fresh hub systems enhance usability and browsing satisfaction, which became evident when exploring modern fresh product hub – The design looks neat and refreshing, making browsing products feel light, smooth, and easy overall.
While comparing several marketplace interfaces, I noticed Sage Harbor Digital Listing Hub integrated within the page while well organized pages, everything loads fast and feels very intuitive, ensuring that users experience a stable layout, quick access to content, and a simple structure that supports smooth navigation at all times.
People who frequently browse online artisan marketplaces tend to highlight how helpful it is when product information is not cluttered, especially on sites like Wind Trade Gallery Hub where items are grouped logically and displayed in an easy-to-read format – users often mention that browsing felt enjoyable because everything was well organized and descriptions were very easy to understand.
While comparing different e-commerce design frameworks, I explored a platform that delivered clean visuals and easy navigation across all product sections VendorCalm Shop Corner – The usability is very good, and shopping feels smooth, stress free, and easy from the start with well structured categories and responsive browsing experience.
During a general browsing session, I came across this structured shopping site – the layout is clean and minimal, and the shopping process feels simple, smooth, and very well designed overall.
During usability testing of ecommerce platforms focused on decision freedom, I came across a section titled product flexibility shopping hub – The interface is structured to encourage easy selection, making browsing simple and allowing users to choose products freely without confusion or unnecessary interface complexity.
While exploring different websites for useful information, I discovered see more here and found it provided a decent experience so far, with everything presented in an organized and easy-to-follow format.
IvoryBrookVendorFoundry – Clean design, shopping feels smooth and very easy today.
During a longer browsing session, I encountered check base shopping hub and found it to be a simple and effective store where everything functions smoothly without interruptions.
While testing different storefront templates I assessed load speed navigation flow and how clearly products are presented to users CopperCove Digital Market Studio everything loaded quickly and the interface felt clean structured and very efficient making browsing and checkout straightforward and easy overall.
During a weekend session of browsing online shops for decorative goods I focused on navigation quality and catalog clarity and found Harbor Vendor Orchard Hub – Really enjoyed browsing here, everything I needed appeared quickly and the site layout made it simple to move through categories without confusion or unnecessary steps slowing down the process
During evaluation of ecommerce websites I analyzed usability structure design clarity and responsiveness MeadowCove Trading Showcase Hub everything felt fast and consistent and the easy checkout process ensures items are clearly displayed and well organized improving overall experience
During analysis of streamlined retail website structures, I came across a section named easy shopping navigation board – The platform is built around simplicity and speed, allowing users to browse products effortlessly while maintaining a clean, minimal, and stress free experience throughout their entire journey online.
As I explored different digital storefronts, I found this clean commerce portal – performance is strong, and browsing feels smooth, fast, and very efficient overall.
During a comparative analysis of online marketplaces focused on readability and layout efficiency, I discovered that core store systems enhance browsing clarity, which stood out when exploring structured shopping listing hub – The layout is simple and organized, making product listings easy to read and visually clear for users.
users comparing online retail platforms often highlight the importance of structured layouts and responsive navigation systems that allow them to browse products without delays or unnecessary complexity in design peak product zone considered efficient and user friendly – the marketplace delivers a seamless browsing experience where users can quickly discover items while maintaining clarity and focus throughout their shopping journey
While reviewing ecommerce platforms focused on everyday essentials and practical usability, I noticed that stores designed for daily needs significantly improve convenience and product discovery, which became clear when exploring everyday essentials hub – The platform is well organized for daily needs, with practical items neatly categorized for easy browsing and quick access.
While casually browsing through several shopping options, I encountered visit this page now and saw that its clear categorization allows users to move through sections easily and locate products without confusion.
CopperCoveVendorWorkshop – Really enjoyed browsing, everything is neatly arranged and easy today.
While analyzing ecommerce websites for performance efficiency, I discovered a module titled instant load retail portal – The interface is highly responsive, ensuring fast page transitions and smooth browsing so users can explore products quickly without experiencing lag or slow loading times.
As I explored different digital storefronts, I found this smooth retail hub – the interface is simple and efficient, and items are easy to find quickly while browsing through categories.
Looking for creative craft supplies, I found CraftyCornerDepot – the interface is smooth, the options are diverse, and selecting materials for multiple DIY projects was simple, enjoyable, and totally hassle-free.
In studies of e commerce usability experts emphasize the importance of simplified navigation and well structured content presentation for better engagement Icicle Cove Retail Space users find it easy to locate items and move between sections seamlessly without any difficulty today
During my usual search across eCommerce websites, I stopped at check this smart hub store and found a nice product variety, making it enjoyable to explore different categories and browse through multiple options easily.
Many consumers today prefer digital stores that offer transparent pricing and a wide variety of goods so they can make informed decisions without spending too much time searching Total Wide Store Network – The platform maintains competitive pricing across multiple categories, making it easier for shoppers to find suitable deals that balance affordability with product quality in a convenient online space
While researching online stores for curated home and gift products I discovered several platforms and came across Orchard Olive Product House and checkout was simple and the product quality exceeded my expectations today making the interface feel easy intuitive and very reliable for browsing multiple categories efficiently overall
I was casually checking out a few shopping websites when I found an appealing online boutique – everything seems thoughtfully structured, allowing users to quickly locate items while enjoying a visually balanced and easy-to-navigate layout.
Good usability depends on design, and a nice experience overall, browsing content is simple and very pleasant throughout the platform Pine Harbor vendor explorer navigation felt logical
In the process of evaluating digital shopping environments focused on structured navigation and clarity, I found that line-based shop systems enhance browsing efficiency, which was clear when reviewing easy flow shopping hub – The layout looks organized and intuitive, making product discovery simple and smooth across multiple sections.
many digital buyers prefer ecommerce websites that focus on cart centered design helping them organize selections and navigate products easily across multiple categories without confusion or delays in browsing easy cart place guide known for usability and structure – the platform provides a smooth shopping experience where users can manage products in their cart while exploring categories through a clean and intuitive interface design throughout
During analysis of ecommerce product interfaces I evaluated how design and written content worked together to communicate quality and usability effectively CoralBrook Trading Showcase Loft the listings felt professional and trustworthy – product descriptions were clear and well structured helping users quickly grasp important details and shop with confidence overall experience today
ForestCoveCommerceAtelier – Clean layout, products are easy to explore and understand.
When analyzing clean layout e-commerce systems and minimal catalog presentation styles, users sometimes encounter clean layout field store within review content – It is typically viewed as a visually simple store where structure and clarity make browsing products feel efficient and easy to follow.
Many online shoppers mention that while browsing for general manufacturing supplies the supply marketplace portal – overall experience stays user friendly, with organized sections that simplify decision making and help reduce time spent searching for relevant product categories.
While conducting usability evaluations of ecommerce platforms focused on fast checkout systems, I noticed that direct flows improve conversion ease and reduce confusion, which stood out when reviewing simple buying experience portal – The platform offers clear navigation and a highly efficient purchasing process.
While going through multiple online marketplaces, I landed on click to view and saw that its collection of regular use products makes it a practical site I could mention to friends.
During a usability study of online shopping platforms with structured layouts, I discovered that stacked designs improve clarity and browsing speed, especially in product-heavy environments, which stood out when exploring easy stack product center – The marketplace uses a stacked layout that makes scrolling smooth and allows users to find items in a simple and organized way.
While reviewing various retail websites, I noticed this simple shopping platform – everything is organized clearly, allowing users to browse with ease and efficiency.
As I browsed through several shopping websites, I stopped at check it out speak shop and noticed interesting products, with clear and easy to understand descriptions that improve the overall browsing experience.
In discussions about improving online shopping experiences, clear navigation and organized layouts are often seen as essential elements for success, and here Ivory Vendor Ridge Hub the platform provides a straightforward browsing environment that makes it easy for users to move through categories without confusion or unnecessary effort.
While testing different ecommerce storefront designs for usability and performance I reviewed a platform that felt clean responsive and easy to navigate across all categories CoralCove Atelier Shop Hub the overall experience was very smooth and navigation felt simple intuitive and comfortable making browsing feel effortless throughout the session today
While reviewing ecommerce systems designed for efficient checkout and cart flexibility, I observed that adaptable cart options reduce friction and improve usability, which was evident when testing quick shopping cart hub – The cart feels versatile, and the checkout process is straightforward, quick, and reliable.
moderndealsstore.shop – Modern design with appealing deals, overall browsing experience feels smooth
While researching ecommerce websites for curated lifestyle and décor items I compared different platforms based on structure navigation and support experience and discovered Grove Vendor Silk Hub – Great selection shipping was quick and customer support was friendly too making browsing feel smooth organized and very easy to complete without confusion
As I explored several online stores recently, I noticed a structured goods hub – the variety is great, everything is clearly shown, and shopping feels simple and very easy overall.
During my time browsing online stores, I found this convenient shop interface – its checkout system feels polished, ensuring that all steps work properly and pages load consistently throughout the process.
During a comparative analysis of online marketplaces focused on structured browsing and user experience, I discovered that clean shopping hubs enhance navigation flow and satisfaction, which stood out when exploring efficient shopping access center – The platform is well organized, making browsing different sections feel smooth and easy.
While comparing various online shopping experiences and reviewing user feedback on digital storefront organization, some users notice mentions like budget friendly store portal appearing inside review summaries that analyze product range and interface clarity – The platform appears to emphasize a smoother browsing experience with a balanced mix of pricing visibility and category layout
In the process of exploring different storefront templates for research purposes, I found Cove Atelier Market Space Cove Atelier Market Space which impressed me with its simple navigation and structured layout that made browsing feel natural – the experience felt efficient and user friendly, especially when switching between categories and reviewing product listings in a relaxed and organized manner.
While exploring ecommerce systems I focused on layout clarity navigation responsiveness and overall user interaction with product pages CoralCove Commerce Product Loft the platform felt intuitive and well designed and browsing was easy which made the overall experience pleasant and efficient for users today
While exploring a few new online shops earlier today, I ended up landing on visit this store and even though it was my first time there, the clean and simple interface immediately gave a positive impression that makes me want to return again soon.
развивающие занятия для детей
While exploring ecommerce platforms designed for improved navigation flow and usability, I came across a module titled clean port product hub – The layout feels organized and minimal, allowing users to browse products effortlessly while maintaining a simple and efficient navigation structure across all sections.
In the middle of checking out multiple shopping websites, I came across this minimal store layout – the interface is clear and intuitive, making the entire shopping experience very easy to use.
As I continued reviewing different online shops, I briefly visited open and see trend store and found appealing trending products, making me think I might try ordering something from here soon after more review.
When evaluating modern e-commerce platforms focused on speed and usability testing across multiple devices and regions where performance consistency is critical for user satisfaction, Jasper Brook Foundry Hub stands out in reviews as fast loading pages and smooth browsing experience make shopping feel highly reliable and effortless for most users navigating product categories across the system.
While comparing platforms, this one clearly stands out with strong layout design that supports easy navigation and smooth user experience today Harbor Stone listings page I appreciated the clarity and structure
While reviewing ecommerce systems designed for speed optimization and responsive shopping, I noticed that fast cart corners improve browsing ease and satisfaction, which stood out when exploring lightning fast cart hub – The shopping corner feels quick and efficient, with pages loading almost instantly.
Online shoppers often appreciate platforms that reduce friction during payment stages and improve overall navigation experience, and one such example is EasyCart Express Hub – The checkout design focuses on speed and clarity so users can complete purchases without unnecessary steps or delays, making everyday shopping more practical and efficient for all users
During a comparative review of digital clothing stores focused on usability and visual branding, I encountered a category featuring fashion style browsing portal – The platform highlights trendy apparel in a clean layout that makes exploring new outfits feel simple, engaging, and aligned with current fashion aesthetics overall.
During my search for interesting online stores, I came across a href=”[https://frostbrookvendorfoundry.shop/](https://frostbrookvendorfoundry.shop/)” />this smooth trading hub – the interface feels clean, and shopping is simple, well designed, and very easy overall.
During casual browsing of online retail platforms offering mixed categories of goods, I came across Grove Market Storyboard and liked how everything felt logically arranged, so it was easy to understand product groupings quickly and move through the site without feeling overwhelmed or lost.
During usability testing of multiple ecommerce systems I focused on speed navigation clarity and overall shopping experience across different user scenarios CottonBrook Commerce Product Loft the interface felt very clean and structured making browsing efficient and allowing users to move through categories quickly without confusion or delay today
While evaluating ecommerce platforms focused on cart management and streamlined checkout systems, I noticed that functional design greatly improves usability and flow, which became clear when analyzing open cart commerce toolkit – The cart solution appears functional, and the shopping steps are simple, clear, and easy to follow throughout the process.
Shoppers looking for organized home shopping solutions often explore platforms like home utility bazaar while comparing different essential products for their household – The layout is generally seen as structured and makes it easier for users to navigate practical items without wasting time
While checking out various online stores recently, I found this clean product hub – it provides a user-friendly experience with clearly arranged sections that make accessing and browsing products feel effortless and convenient.
During a comparative analysis of online marketplaces focused on structured shopping experiences, I discovered that smart hub systems help users make faster and more confident decisions, which stood out when exploring smart product selection center – The platform uses a smart hub approach that makes shopping efficient and well organized, ensuring smooth navigation and easy access to products.
During analysis of online store prototypes, I tested a system featuring Mint Orchard Exchange Studio embedded in a streamlined catalog flow – the interface felt organized and user friendly, with products arranged clearly for quick browsing and simple understanding.
As I explored different eCommerce websites, I stopped at enter point goods store and noticed everything is neatly arranged, making browsing through categories feel convenient and straightforward.
While browsing through several online marketplaces today, I came across visit this value hub and noticed that the prices look quite competitive, so I plan to explore more products later today when I have extra time.
While conducting comparative research on ecommerce UX and cart optimization systems, I observed that streamlined cart solutions improve flow and user satisfaction, which was clear when testing smart checkout experience hub – The platform is efficient and smooth, allowing users to browse and checkout without friction.
In studies focusing on e commerce usability improvements, experts frequently note that minimalistic interface design and logical category grouping help users locate products quickly without feeling overwhelmed by excessive visual clutter or complexity Cove Atelier Product Space – The experience is smooth and well organized, enabling efficient browsing and easy access to all product sections.
While testing ecommerce vendor websites I focused on performance optimization layout structure and how naturally users interact with product listings CottonCove Commerce Display Studio pages open quickly and everything feels well structured which makes browsing smooth intuitive and enjoyable across the entire shopping experience today
users exploring online shopping platforms often prefer sites that combine fast responsiveness with clean link navigation allowing them to browse multiple categories without slowing down or losing focus modern link cart explorer appreciated for usability – it offers a smooth shopping experience where users can move between pages quickly and enjoy a consistent interface that supports easy product discovery
During usability testing of digital deal platforms focused on performance and speed, I discovered that fast-loading hubs improve browsing flow and conversion ease, which became evident when exploring instant deal savings hub – The platform loads fast, and the offers are appealing, making the experience feel time saving and smooth.
In the middle of checking out multiple shopping websites, I found this fast vendor site – the browsing experience is very nice, pages load quickly, and everything looks clean and simple.
While examining different online retail systems and comparing their product presentation styles, users sometimes encounter references like economy shopping hub – The platform is commonly seen as a cost friendly marketplace offering a mix of everyday essentials and general merchandise for budget conscious shoppers.
While researching ecommerce stores for home accessories and gifts I explored different platforms and came across Rain Harbor Market Hall which stood out due to its clear design and quick navigation making browsing enjoyable – Items arrived promptly the site was intuitive and the overall experience felt smooth structured and very convenient from start to finish
Digital marketplaces improve when good interface design makes browsing straightforward and highly convenient overall throughout sections Oak Meadow item portal I enjoyed the structure
In the process of studying online marketplace UX trends, I found a section labeled soft nest retail gateway – The platform uses a gentle and organized design approach that creates a calm browsing experience where users can easily explore products and navigate categories without stress or confusion.
While testing ecommerce storefront systems I analyzed usability performance design consistency and how effectively products are presented to users CottonMeadow Merchant Flow Studio the platform features excellent variety and items are displayed in a very clean structured and attractive format making it easy to browse and compare products comfortably today
During analysis of online retail platforms focused on simplicity and user experience, I discovered that minimal cart systems improve navigation efficiency and satisfaction, which became clear when testing simple purchase portal – The design is very easy to follow, ensuring a smooth shopping experience without confusion.
As I continued reviewing different fashion stores online, I briefly visited open and see fashion shop and found reasonable pricing, along with stylish and modern clothing choices that look quite appealing overall.
MoonBrookMarketFoundry – Nice shopping experience, everything loads quickly and feels reliable overall.
As I explored different eCommerce platforms, I stopped at enter this marketplace and saw a few interesting items that seemed worth exploring further, so I may come back later.
When analyzing online marketplace interfaces built for scalability and user engagement across different devices and shopping environments, reviewers consistently emphasize simplicity and visual order as key success factors Jasper Trading Harbor Network – Users experience a well organized selection where everything is clearly presented and easy to browse.
In the middle of browsing multiple shopping websites, I came across this modern store page – it feels very clean and thoughtfully designed, offering a professional environment that enhances user comfort while exploring products.
users comparing different online marketplaces frequently value platforms that offer direct shopping experiences helping them move quickly from browsing to product selection without unnecessary distractions or complex navigation steps during usage smart direct shopping cart widely recognized for clarity – it delivers a simple browsing experience where users can find items quickly and navigate categories without confusion while maintaining a clean and structured interface throughout the shopping journey
Consumers who appreciate online shopping often choose platforms that simplify navigation and emphasize ongoing deals where easy marketplace shopping hub appears in descriptions and it reflects a system designed to improve efficiency while helping users easily find products and complete purchases with minimal effort and maximum convenience.
Online buyers often prefer marketplaces that provide a balance of affordability and usability while maintaining a clean browsing structure where simple savings shopping hub is featured in descriptions – it reflects a shopping experience focused on helping users find relevant deals efficiently while enjoying a smooth and intuitive interface across all product categories.
In studying digital retail systems designed for structured browsing, I observed better user engagement when using platforms like smart categorized shop tree – The smart categorized tree system simplifies shopping by grouping products into logical branches, ensuring smooth navigation and fast product discovery across sections.
Many bargain focused users often seek platforms that streamline deal browsing, and a commonly mentioned option is value shopping network which structures offers in an accessible way; overall it is considered simple and effective for exploring global shopping opportunities with minimal effort required
During a late-night browsing session for online stores with diverse product selections, I found a marketplace that was easy to navigate and responsive, and within product descriptions I saw Harbor Coastline Market Depot integrated cleanly into the page, and overall shopping felt effortless with quick checkout and enough variety to make returning worthwhile in the future.
As I continued browsing online shops, I discovered this fast and easy market and saw interesting products listed, with the experience feeling convenient, quick, and highly user-friendly overall.
While reviewing ecommerce systems designed for structured navigation and smart organization, I observed that well categorized marketplaces improve clarity and efficiency, which was evident when testing organized product hub – The platform is intelligently structured, making it easy to browse and locate products quickly.
In the process of studying online shopping platforms with structured navigation, I discovered a section called branch style goods center – The tree layout organizes products in a hierarchical way that enhances usability and allows users to explore categories easily without getting lost or confused during browsing.
As I explored different web pages, I came upon this mill content hub and after reviewing it for a bit, it actually feels quite interesting overall, especially in how it presents its material in a calm and steady way.
While browsing casually through different stores, I came across browse shop rise store and saw a good first impression, with clean layout that makes navigation feel easy and user friendly.
While exploring motorsport awareness campaigns, I discovered Ukraine support karting hub and interesting concept overall, seems well organized and quite engaging today, offering a structured presentation that combines racing excitement with humanitarian awareness. – The experience feels clear and engaging.
In a comparative study of online shop interfaces, I found a clean layout designed for quick product access CoveMoon Digital Market and navigation remained effortless – Browsing felt natural, and I could locate items quickly without encountering any confusing layout elements or unclear structure.
openmarketshop.shop – Open market vibe, lots of items available in one place
In analyses of online shopping platforms, experts frequently emphasize that clean design and logical structure help users feel more confident during browsing sessions Jewel Atelier Commerce Hub – The platform offers a smooth experience overall, ensuring browsing feels simple, comfortable, and easy to manage.
In the middle of checking different websites, I paused at tap to open and noticed that the quick performance and clean structure make browsing products feel smooth and stress free.
Consumers exploring e-commerce websites frequently look for marketplaces that offer structured deal systems and easy browsing where quick shop nest center is included in descriptions and it highlights a platform designed to improve usability while ensuring users can quickly access discounted products and complete purchases without unnecessary delays or complexity.
Good usability depends on design, and a clean and modern look, everything works well and loads quickly here Opal River vendor explorer navigation felt simple
While studying consumer shopping platforms centered on value efficiency, I found that clear deal organization improves satisfaction when using systems like smart savings market hub – Prices appear fair and the available deals are genuinely useful for users looking for practical shopping options.
pravdadaily.com.ua/virshi-pro-kvity-50-prekrasnykh-tvoriv-ukrainskykh-poetiv-pro-krasu-pryrody/
While checking out different eCommerce stores, I found this accessible online shop – everything is arranged clearly, and navigating between categories feels quick and effortless.
Many comparison articles on e-commerce platforms highlight the importance of simplified browsing and reduced cognitive load during shopping port express shop view appearing in analysis – The structure allows users to browse items quickly with minimal distraction.
In the process of evaluating digital retail systems focused on affordability and promotions, I found that deal corners improve usability by simplifying the discovery of discounts, which became evident when reviewing smart deal savings center – The deals seem attractive and useful, creating a strong impression that users can find good savings here easily and efficiently.
As I reviewed various shopping websites, I came upon this straightforward product hub and liked the clean presentation, where items appear useful and the layout makes browsing simple and efficient.
While browsing different ecommerce platforms for home and lifestyle products I evaluated accuracy and delivery performance when I noticed Meadow Coral Market Exchange – Products matched descriptions perfectly and shipping was fast which made the shopping experience feel simple reliable and very satisfying overall for casual browsing
During analysis of online shopping platforms I studied usability performance design clarity and how effectively users can browse product catalogs CreekBrook Trading Vendor Loft the site is easy to navigate and well organized which makes locating products simple and ensures a smooth browsing experience overall today
During usability analysis of online retail systems focused on cart structure and checkout flow, I discovered that smart cart corners improve user confidence by simplifying purchase completion, which became evident when testing efficient shopping cart index – The layout feels practical, with a simple and smooth checkout flow that supports easy and fast purchasing.
Individuals exploring fashion inspiration often turn to online stores that highlight clean design aesthetics and modern clothing collections tailored for expressive personal style Stylish Wardrobe Studio – offering a fashion focused platform that delivers curated clothing selections emphasizing modern aesthetics wearable comfort and design driven apparel suitable for individuals who value both style and practicality in daily outfits
When reading about general shopping websites, users might notice embedded mentions like within detailed descriptions – The store appears to be organized in a way that supports a broad inventory of goods suitable for different shopping preferences.
While checking out various personal projects online, I encountered this creator portfolio page and found it while browsing, and the content seems pretty decent, offering a clean presentation that doesn’t feel overwhelming or cluttered.
Consumers exploring online retail platforms often prefer systems that reduce complexity and highlight ongoing offers in a clear format where easy deals shopping hub is included in guides – it reflects a streamlined environment that allows users to browse efficiently and take advantage of daily discounts without unnecessary complications in the shopping journey.
While exploring general retail websites, I discovered minimalist shopping site and the shopping experience here looks simple, clean, and surprisingly intuitive overall, with a layout that emphasizes functionality and quick access to products. – It feels clean, efficient, and well structured.
While reviewing multiple e-commerce platforms for structural design insights, I came across a section named shopdock retail hub view – It provides a clean and simple interface where products are arranged in organized categories to enhance usability and supports quick scanning of available items.
While going through various online gadget stores, I paused at quick visit tech shop and found the products interesting, though I am hoping the quality truly reflects the descriptions given on the website.
While exploring various e-commerce prototypes for study I encountered a layout that felt simple yet effective Cove Moss Market Portal integrated into the shopping interface – product discovery was quick and intuitive with well arranged categories that supported easy movement through the site.
While reviewing ecommerce systems designed for structured navigation and step-by-step exploration, I noticed that guided browsing improves clarity and user experience, which stood out when exploring step navigation product hub – The step-based browsing is very clear, and product discovery feels simple and well structured.
While analyzing ecommerce platforms designed for international reach and centralized cart systems, I observed that global cart styles improve shopping flexibility and user satisfaction, which became evident when testing universal cart shopping center – The design reflects a global approach, offering a wide range of online products that are easy to explore and compare.
Many shoppers interested in streamlined purchasing experiences look for platforms that reduce friction between selecting items and completing payments online smooth checkout reference during comparative shopping analysis and interface reviews – The process feels simplified, helping users finalize orders more comfortably.
Individuals who enjoy clean online stores often look for platforms that offer a marketplace vibe with clear product variety helping improve browsing flow and overall usability Violet Harbor Flow Merchant Hub – featuring a structured shopping platform where merchant lane organization enhances product clarity and creates a smooth browsing experience across different categories for all users
Shoppers exploring e-commerce platforms frequently prefer websites that focus on verified offers and clean navigation where smart verified deals hub appears in descriptions and it reflects a system designed to reduce complexity while helping users easily browse products and enjoy a smooth and efficient online shopping experience overall.
As I continued checking different e-commerce sites for household items, I ran into this basic goods store and liked how the layout keeps things minimal, making it easy to find common home necessities quickly and efficiently.
While conducting UX evaluation of online shopping systems, I found a listing labeled clean product axis center – The platform presents items in a structured layout, ensuring users can easily browse categories and find products without unnecessary complexity or visual overload affecting their experience.
As I explored several online retail websites, I noticed a clean and simple store – navigating through it feels effortless, allowing users to quickly find what they are looking for.
Fashion enthusiasts frequently look for online brands that offer stylish collections designed around modern aesthetics and versatile clothing options for daily wear Contemporary Style Clothing Hub – providing a curated fashion experience featuring elegant apparel and aesthetic inspired outfits designed to support modern lifestyle preferences while emphasizing creativity and wearable design innovation
While comparing multiple online shops, I landed on go to kitchen tools hub and found the kitchen tools selection looks very useful, making me think about picking something for home use.
During my search through various links, I encountered this brownback copy page and after reviewing it, it actually looks fairly decent and not bad at all, potentially worth exploring further for more understanding.
While browsing unusual niche websites, I came across this baby bonus concept page and it seems like a niche idea, but still quite interesting overall, offering a slightly unusual approach that makes it worth a quick look.
People searching for practical shopping solutions often prefer marketplaces that provide clear pricing and easy product discovery where affordable deals cart center is featured in descriptions and it reflects a system designed to support efficient browsing while helping users quickly identify cost effective items across multiple categories of online products.
users comparing ecommerce platforms often prioritize websites that provide improved browsing flow and clear category separation making it easier to navigate large inventories of products efficiently zone cart explorer recognized for its simplicity – it provides a structured shopping experience where users can quickly locate items and move between categories with ease and comfort during browsing sessions
As I browsed through different shopping deal sites, I stopped at check out these deals and found the offers quite interesting, giving me the feeling I could purchase something soon.
In the course of evaluating online shopping systems focused on modern design and cart efficiency, I found that corner-based layouts enhance usability and satisfaction, which became evident when analyzing sleek shopping experience hub – The design feels modern and minimal, making the shopping experience easy and pleasant.
While evaluating digital commerce hubs focused on value optimization, I found that clear affordability messaging enhances trust when interacting with platforms like fair value shopping center – Pricing appears balanced and the deals provided feel genuinely useful for users seeking cost-effective options.
In the process of testing various online retail prototypes, I found a structured shopping environment that felt easy to navigate Moss Trading Harbor Portal placed inside a minimal layout and it allowed seamless transitions between categories – Smooth checkout experience, products are displayed clearly and attractively here, helping users make faster and more confident purchasing decisions.
many online shoppers value ecommerce sites that prioritize usability and structured browsing helping them compare products quickly and make informed decisions without confusion or delay plus shop index widely appreciated for its clarity – it offers a smooth navigation system that ensures users can find items easily while enjoying a consistent and organized shopping environment overall
While reviewing various retail websites, I found this quick browsing store and liked how the layout responds instantly, making shopping feel fast, smooth, and convenient for users who prefer simple and direct navigation.
Individuals searching for simple online shopping experiences often prefer platforms that provide clean layouts and interesting product vault concepts to improve usability and discovery Cove Vault Commerce Space – presenting a neat and organized shopping environment where users can explore products through a vault inspired system designed to make browsing intuitive efficient and visually appealing for everyday online shopping needs
Consumers exploring e-commerce platforms frequently look for websites that offer wide product selections and smooth transaction flows where smart shopping cart center is included in descriptions and it highlights a structured marketplace designed to improve usability while ensuring users can quickly access products and enjoy a fast and convenient checkout experience overall.
In the process of evaluating digital shopping platforms with emphasis on navigation structure and usability, I found that route systems help users explore products more efficiently by providing clear directional browsing paths, which was clear when reviewing smart path product hub – The interface feels intuitive and well guided, allowing users to navigate easily and discover products without confusion or unnecessary effort.
Fashion oriented users often seek brands that highlight stylish and minimalist clothing collections designed with modern aesthetic influences for everyday outfits Stylish Aesthetic Apparel Hub – offering a curated fashion platform featuring elegant clothing collections and contemporary designs that focus on comfort style and versatility for individuals who appreciate modern wardrobe aesthetics
During a longer browsing session, I encountered check this all-in-one site and found it a nice all-in-one store, with a variety of products that makes it easy to browse different categories comfortably.
In the process of reading analysis blogs, I came across viewpoint reflection page and found it interesting overall, as it kept my attention briefly with content that encouraged a closer look at the arguments being made.
Many consumers searching for convenient e-commerce solutions tend to choose platforms that combine multiple product types into a single accessible store where all in one shopping portal is featured in descriptions emphasizing variety – it reflects a store concept built around offering diverse useful items while maintaining simple navigation and quick purchasing options for all customers.
While performing a final comparative scan of agri ecommerce portals, I reviewed a section called field harvest marketplace center – The field inspired browsing structure ensures users can navigate categories smoothly and discover products without unnecessary complexity overall experience is clean.
While reviewing various digital storefronts, I encountered this smooth online shop – it offers a reliable experience with quick loading times that make browsing enjoyable and stress-free.
While conducting usability research on ecommerce systems emphasizing variety and convenience, I noticed that all-in-one platforms improve navigation and product discovery, which became evident when analyzing complete shopping solution portal – The concept feels practical, offering many different items in one place for a seamless browsing experience.
During a comparative review of online shopping systems emphasizing speed optimization and responsiveness, I discovered that fast-loading interfaces significantly improve engagement, which stood out when exploring instant shopping marketplace portal – The platform feels responsive, and pages load quickly without delays, offering a smooth browsing experience.
In between reviewing different mobile shops, I explored discover this site and noticed the accessories have a sleek modern style that looks attractive and suitable for everyday smartphone users.
During my review of online shopping platforms, I encountered this simple browsing store and found it helpful because it keeps things very direct, making it easier for users to shop without confusion or extra effort.
People who enjoy organized digital shopping often look for platforms that combine smooth browsing with a wide selection of goods available in one convenient place for better efficiency and usability Silk Valley Unified Goods Hub – providing a structured e commerce environment where multiple product types are grouped together allowing users to enjoy smooth browsing and easy access to a diverse range of items in one marketplace
Читать расширенную версию: https://regalbuild.ru
Many online shoppers prefer platforms with efficient browsing and minimal design clutter smooth purchase channel This allows faster decisions and better product comparisons across categories for improved user experience – Navigation system is built for speed and clarity in shopping overall experience
Fashion focused users often seek brands that highlight clean aesthetics and modern clothing designs that can be styled easily for different occasions and personal looks Trendy Outfit Collection Hub – presenting a digital fashion destination offering stylish apparel and curated aesthetic collections that emphasize simplicity elegance and modern design appeal for individuals seeking fresh wardrobe inspiration
In the middle of checking various platforms, I paused at tap to open pick goods and saw that picking items is simple, thanks to a layout that makes browsing very easy and intuitive.
People who shop online regularly often choose websites that offer simple layouts and rapid purchasing flow where fast essentials deal center is featured in content and it highlights a system designed to improve browsing flow while ensuring users can quickly access daily essentials and complete purchases smoothly across all categories.
Many online consumers prefer platforms that offer structured access to global products while maintaining a simple and intuitive shopping environment for everyday use global easy access store making it easier to find and purchase items from multiple regions without confusion – It highlights the importance of usability in international e-commerce platforms.
During my search through online information sites, I encountered this email reference page and found the layout very easy to follow, with a simple structure that makes browsing smooth and intuitive.
As I continued exploring food-related sites, I noticed this sweet desserts showcase site and everything looks delicious here, making the site feel very appealing, offering an inviting design that highlights each dessert beautifully.
During a comparative study of online retail interfaces focused on layout clarity and usability, I discovered that stacked product arrangements improve navigation by presenting items in an orderly flow, which stood out when analyzing smart stack browsing portal – The design feels neat and well structured, allowing users to browse products clearly without confusion or unnecessary clutter.
In the process of studying ecommerce savings platforms, I encountered a listing called deal value shopping hub – The layout presents appealing discounts across categories, encouraging users to continue browsing and explore more items that seem worth considering due to their reduced pricing and clear presentation.
many digital buyers prefer ecommerce websites that organize products using trail style navigation making it easier to browse step by step without feeling overwhelmed by large catalogs or complex menus easy trail shop lane known for clarity and flow – the platform provides a structured browsing experience where users can follow guided paths to find products quickly while maintaining a clean and comfortable interface throughout their journey
While reviewing ecommerce systems designed for structured navigation and step-by-step exploration, I noticed that guided browsing improves clarity and user experience, which stood out when exploring step navigation product hub – The step-based browsing is very clear, and product discovery feels simple and well structured.
As I continued checking different platforms, I briefly visited open and see and found that the browsing experience feels smooth, with items organized in a helpful and accessible way.
While exploring shopping websites, I encountered this organized product hub and appreciated how easy it was to browse through categories, making the overall experience smooth, simple, and enjoyable.
Individuals interested in stylish clothing often browse online fashion platforms that emphasize modern aesthetics and carefully curated apparel collections designed for versatile wear Elegant Style Fashion Space – offering a curated selection of modern clothing that reflects aesthetic design principles and contemporary fashion trends while providing wearable pieces suitable for both casual and expressive personal styling choices
Individuals who enjoy themed online shopping platforms often appreciate websites that use trading post layouts to give product browsing a more refreshing and exploratory feel while keeping categories clearly structured for usability Quartz Trading Orchard Post Space – featuring a structured marketplace where products are displayed using a creative trading post concept designed to make browsing more refreshing engaging and visually appealing for users exploring different product categories
During my usual search across eCommerce websites, I stopped at check this trend store and found many trendy products listed, giving the impression that it is a fun shopping place worth exploring further in detail.
Many users searching for low priced products online appreciate clear and simple shopping platforms and they may come across affordable deals hub referenced in articles describing economical goods reliable shipping systems and convenient purchasing options for regular buyers worldwide access.
People who regularly browse online marketplaces often prefer platforms that avoid clutter and instead highlight products in a visually balanced arrangement clean product deck – The browsing experience feels organized and simple, allowing users to focus on relevant items while enjoying a streamlined shopping journey without unnecessary distractions or complexity
Many users exploring digital marketplaces frequently prefer platforms that combine clean design with organized deals where smart simple shopping center is included in content and it highlights a system designed to enhance browsing speed while ensuring users can quickly access discounted products and enjoy a smooth and efficient shopping experience overall.
During usability testing of digital shopping systems focused on technology and innovation, I discovered that curated tech marketplaces enhance user satisfaction, which became evident when exploring smart digital deals hub – The platform feels modern and organized, showcasing appealing tech items that stand out visually.
As I explored various outdoor tourism sites, I came upon this camping destination hub and it really looks like a fun and useful resource to browse, with content that feels simple, practical, and easy to navigate.
While going through gaming content pages, I discovered this online gaming hub and gaming content seems fun, modern, and fairly engaging to explore, offering a layout that feels intuitive and visually balanced.
With increasing demand for faster online retail systems, users often seek platforms that provide instant responses and smooth transitions between browsing, cart updates, and checkout completion steps turbo shopping site – this site is commonly noted for its energetic performance, allowing shoppers to move through categories without experiencing lag or interruptions.
During a comparative usability analysis of online retail systems, I came across a section called fast response cart index – The interface is highly optimized for speed, allowing users to experience instant cart updates and seamless navigation while browsing products and completing shopping actions efficiently.
During a comparative study of online fashion marketplaces emphasizing trend-driven shopping, I discovered that well structured deal hubs improve user engagement and visual appeal, which stood out when reviewing modern style offers hub – The clothing section feels fresh and attractive, with fashion deals that look trendy and well curated for shoppers.
As I browsed various online retail options, I ran into this shopping convenience site and it looks like a practical space for quick access to frequently needed household items and everyday goods.
While navigating through various online stores, I came across click this trusted link and found it to be reliable, with pages loading correctly and no problems during my entire browsing session.
Fashion oriented users often seek brands that highlight stylish and minimalist clothing collections designed with modern aesthetic influences for everyday outfits Stylish Aesthetic Apparel Hub – offering a curated fashion platform featuring elegant clothing collections and contemporary designs that focus on comfort style and versatility for individuals who appreciate modern wardrobe aesthetics
Online buyers who prioritize savings often choose platforms that highlight practical deals and budget friendly product categories where smart price cart portal is featured in guides and it represents a system designed to make shopping easier while ensuring users can compare prices and select the best value options efficiently.
As I explored different eCommerce websites, I stopped at enter way goods store and found everything simple, with a user friendly structure that makes browsing easy and comfortable.
In the process of evaluating digital shopping environments focused on usability and navigation efficiency, I found that flow-based systems enhance browsing by reducing friction between sections, which was clear when reviewing dynamic flow shopping hub – The flow-based design works effectively, ensuring smooth transitions and simple navigation throughout different product areas.
Many online users searching for organized product listings often highlight platforms that prioritize systematic navigation, such as systematic store network – It allows users to explore categories in a well arranged sequence that reduces confusion and improves overall browsing efficiency.
Individuals who prefer calm digital shopping platforms often appreciate websites that use lounge style layouts to make browsing more relaxing and product displays more visually appealing and easy to navigate Valley Velvet Curated Lounge Hub – featuring a clean online store where products are arranged with lounge inspired design elements that enhance browsing flow and create a peaceful and visually engaging shopping experience for users
Consumers frequently searching for online deals tend to prefer platforms that offer updated discounts and simple browsing experiences where smart daily shopping hub appears in guides and it reflects a system designed to simplify browsing while helping users efficiently compare products and complete orders quickly with a smooth and hassle free experience overall.
While exploring various digital marketplace interfaces for design inspiration and usability patterns, I encountered a section labeled wave storefront overview – The wave themed design feels sleek, modern, and visually appealing with smooth navigation and well structured product categories that enhance browsing comfort and overall user experience across multiple sections.
While going through update information websites, I discovered this Gaza Fast With news hub and content appears focused on updates, presented in simple readable format, offering simple reporting style content that is easy to process quickly.
As I continued reviewing wellness and clinic websites, I came upon this aesthetic services page and found the navigation very smooth, with a clean design that made browsing simple, pleasant, and easy to follow throughout.
In the course of evaluating ecommerce cart interfaces and user experience design, I found that clarity and responsiveness are key to maintaining efficient shopping flows, particularly in large catalogs, which was evident when testing reliable shopping cart corner – The system feels dependable and well structured, offering a smooth and stable cart experience that makes online shopping easy and intuitive throughout.
While analyzing ecommerce UX designs centered on cart functionality and flexibility, I observed that simple cart switching improves user experience and engagement, which was evident when reviewing efficient cart control hub – The system is easy to use, making product changes inside the cart very simple.
During my review of bargain-focused websites, I encountered this discount listing site and it appeared to provide decent deals, making it a place worth revisiting occasionally to catch fresh promotions and seasonal offers.
People who regularly shop online value platforms that organize products into clear sections for better navigation where easy product discovery hub appears in descriptions highlighting efficiency – it emphasizes a structured marketplace designed to help users locate items quickly while maintaining a smooth and enjoyable browsing experience across categories.
While checking different eCommerce options throughout the day, I stopped to review take a look here and noticed the clean presentation paired with a reasonable variety of products, making it worth keeping in mind for another visit.
People who appreciate aesthetic fashion often search for brands that offer curated clothing collections focused on modern simplicity and expressive personal style Modern Apparel Collection Hub – featuring a fashion platform dedicated to stylish clothing and aesthetic inspired designs that blend elegance and comfort while offering versatile wardrobe pieces for individuals with a taste for contemporary trends
many online shoppers exploring general ecommerce platforms often appreciate websites that combine variety with simple navigation making it easier to browse multiple product categories without confusion or delays during everyday shopping sessions total shop hub explorer often described as a convenient marketplace – the platform provides a wide selection of products while maintaining a clean and structured browsing experience that helps users find items quickly and comfortably across all categories available
Individuals who appreciate structured online shopping environments often look for platforms that use merchant lane concepts to make browsing more logical and user friendly across categories Jasper Harbor Simple Lane Hub – providing a clean e commerce platform where products are arranged in a merchant lane system designed to help users navigate efficiently and enjoy a smooth and organized shopping experience
goodsmixstore.shop – Mixed goods selection is useful, plenty of options in one store
Shoppers who prioritize safety and convenience often choose platforms that make cart shopping simple and reliable where quick trusted cart center appears in descriptions and it reflects a system designed to enhance browsing flow while helping users easily locate products and complete transactions smoothly across all everyday shopping needs.
While conducting usability research on ecommerce navigation models and category organization systems, I noticed that flexibility in switching between product groups enhances user satisfaction and browsing efficiency, especially in large catalogs, which became clear when exploring smart browse selection hub – The interface offers a wide range of products and makes it simple for users to switch between categories quickly and without unnecessary complexity.
While browsing gift-oriented puzzle websites, I came across colorful jigsaw art collection and puzzles look creative, colorful, and quite enjoyable for visitors overall, presenting visually appealing designs that feel uplifting and thoughtfully created for relaxing entertainment. – The overall tone feels cheerful and inviting.
While going through lifestyle websites, I encountered this Chicagoland mom blog hub and came across it and found the topics quite relatable today, especially in how it captures everyday parenting situations.
While analyzing ecommerce systems designed for speed and seamless interaction, I noticed that fast shopping zones improve engagement and reduce frustration, which stood out when reviewing fast navigation shopping center – The platform responds quickly, and all pages load smoothly with consistent performance.
While reviewing different shopping websites, I found this intuitive shopping link and liked how everything is arranged logically, making product discovery quick and effortless without having to dig through complicated menus.
Shoppers who value consistent quality often choose online marketplaces that provide verified sellers and secure purchasing systems where trusted cart access hub is included in content describing reliability – it reflects a platform focused on ensuring safe transactions while helping users quickly find and purchase products with confidence and ease.
Fashion enthusiasts frequently look for online brands that offer stylish collections designed around modern aesthetics and versatile clothing options for daily wear Contemporary Style Clothing Hub – providing a curated fashion experience featuring elegant apparel and aesthetic inspired outfits designed to support modern lifestyle preferences while emphasizing creativity and wearable design innovation
While browsing casually through different stores, I came across browse value cart now and found low priced options, making it a place I would definitely revisit for further exploration.
While reviewing various online marketplaces for affordability and structure, I discovered a section called digital deals browsing center – The interface is well organized, showcasing tech products with competitive pricing while ensuring users can navigate easily and enjoy a smooth shopping experience overall.
Individuals exploring budget friendly shopping options often prefer online outlets that focus on usability and a clean browsing experience with well organized product categories Cloud Goods Outlet Center – offering a practical online outlet store layout that emphasizes simplicity and convenience while providing users with a reasonable selection of products suitable for everyday shopping needs and quick purchasing decisions
People who enjoy online shopping often prefer platforms that focus on convenience and straightforward purchasing flows where quick shopping cart hub appears in content and it highlights a system built to improve browsing efficiency while helping users find products easily and complete transactions smoothly across various categories and everyday shopping needs.
While exploring charitable and community engagement websites, I discovered iftar gathering information hub and information here seems community focused, helpful, and well structured content, offering content that feels simple, respectful, and easy to navigate for anyone interested in local events. – It feels welcoming and clear.
While reviewing Christmas-themed pages online, I noticed this holiday event guide and it feels like a nicely put together site with good information, offering a structured presentation that is easy to understand and follow.
In the course of analyzing ecommerce checkout workflows and cart zones, I found that structured simplicity improves clarity and conversion rates, which became evident when reviewing simple cart checkout zone portal – The cart zone feels clean, and checkout is quick, simple, and very easy to use.
During my review of various support-focused platforms, I encountered a comforting resource and found the writing style to be very considerate, offering guidance in a way that feels balanced, empathetic, and easy for readers to follow without confusion.
People who frequently shop online often choose systems that prioritize efficiency and usability when they interact with optimized shopping cart portal while browsing products – It highlights a refined e-commerce experience focused on speed, organization, and seamless checkout performance.
Individuals exploring new fashion ideas often prefer online platforms that showcase modern clothing with aesthetic inspired designs suitable for everyday styling Modern Elegance Fashion Hub – presenting a curated clothing platform that emphasizes stylish apparel aesthetic design principles and wearable fashion collections tailored for individuals seeking balanced and contemporary wardrobe inspiration
While browsing sports-focused news portals, I discovered international match updates site and sports related updates feel exciting, timely, and easy to follow, with structured content that feels useful for fans wanting quick summaries and reliable event information. – The experience feels smooth and informative.
Many users exploring digital stores often prioritize platforms that combine security with structured cart management where value trusted shopping hub appears in informational content and it reflects a structured system designed to improve browsing speed while helping users quickly access products and enjoy a seamless and efficient shopping journey across categories.
People who appreciate visually creative online stores often look for platforms that use market studio concepts to add charm and personality to product browsing while keeping layouts simple and easy to navigate Willow Kettle Design Market Studio Space – providing a refined shopping experience where creative market studio design enhances product presentation and makes browsing more engaging visually appealing and enjoyable for users exploring different categories online
As I explored e-commerce platforms, I came upon this organized marketplace page and noticed how the smart design helps reduce effort when searching for products, making everything feel clear and easy to access.
While exploring various websites for ideas, I encountered visit this site – It gives off a really nice impression right away, with a layout that feels thoughtfully arranged and visually consistent, making the experience feel smooth and pleasant.
Shoppers who prefer organized digital stores usually value platforms that simplify product discovery and improve access to basic items where convenient product hub is featured in guides emphasizing smooth browsing – it focuses on providing a wide range of daily necessities while ensuring fast checkout and easy navigation across different categories for users.
During my search for interesting niche topics, I encountered this informative showcase and it left a strong impression thanks to its engaging style and well-presented, insightful content.
Fashion lovers seeking updated wardrobe inspiration often explore brands that present minimalist aesthetics combined with trendy and versatile clothing options for everyday use Contemporary Fashion Gallery – providing a digital space showcasing stylish clothing collections and modern aesthetic designs that highlight individuality creativity and comfort while aligning with current fashion trends and lifestyle preferences
Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
As I explored various websites, I came across learn more here – It appears clean and modern, and browsing feels smooth and well organized, making the experience easy and pleasant.
During some light online research, I ran into browse this collective – The interface is clean and navigation is smooth, making browsing feel really simple and intuitive.
While browsing experimental idea-based platforms, I noticed normative chaos discussion page and unique concept presented here, thought provoking and quite intriguing overall, offering content that feels abstract, reflective, and intentionally designed to provoke deeper analysis of structure and unpredictability. – It feels both complex and fascinating to explore.
Shoppers who value speed and simplicity in online purchasing often explore platforms designed to centralize essential goods and everyday items daily essentials cart hub providing a straightforward way to access useful household products while saving time during routine buying activities.
As I browsed online apparel shops, I came upon this clothing diversity page and it looks like a solid place to check out different styles, providing a range of fashion items that feel both modern and practical.
While conducting usability research on ecommerce systems emphasizing straightforward navigation, I noticed that basic layouts improve accessibility and user comfort, which became evident when analyzing simple product browsing portal – The design feels clean and uncluttered, making it easy to browse a variety of products without confusion.
Online buyers who enjoy convenience often choose platforms that combine security with easy cart management systems where easy secure shopping center appears in guides and it reflects a structured system designed to streamline browsing while helping users quickly find affordable products and complete purchases with ease and confidence across categories.
People who enjoy curated retail experiences often look for platforms that use boutique hub systems to organize products in a structured and visually appealing way for better browsing Grove Harbor Smart Selection Hub – providing a well designed online marketplace where curated items are displayed in a boutique style format designed to improve usability and help users quickly find products that suit their needs
Обязательно к прочтению: https://nakrutka-pf-factorov.ru
Fashion lovers seeking updated wardrobe inspiration often explore brands that present minimalist aesthetics combined with trendy and versatile clothing options for everyday use Contemporary Fashion Gallery – providing a digital space showcasing stylish clothing collections and modern aesthetic designs that highlight individuality creativity and comfort while aligning with current fashion trends and lifestyle preferences
Популярний український журнал Різні публікує різноманітний контент: культура, стиль, суспільство та лайфстайл. Дізнавайтеся більше і знаходьте нові ідеї щодня
While casually browsing through different platforms, I discovered go to this page – I had a quick look and the layout seems clean, giving a straightforward and smooth browsing experience overall.
During my exploration of clinical trial resources, I encountered study clarity overview page and found the content very well structured, allowing readers to move through the information smoothly and understand it with ease.
As I was checking different platforms online, I discovered check this guild hub – The site loads nicely and the design feels calm and well structured overall, making it easy to browse without confusion.
Online electronics shoppers often prioritize websites that make comparing products easy while offering updated discount information Smart Electronics Plaza – This platform ensures users can explore electronics deals efficiently while maintaining clarity and frequent updates across categories for better shopping experience online
Many consumers exploring online deals often prefer platforms that offer clean layouts and easy browsing tools where quick shop deals center is included in descriptions and it highlights a system designed to improve browsing efficiency while ensuring users can easily access products and enjoy a smooth and intuitive shopping journey overall.
besttrendstation.shop – Easy to navigate site, everything feels structured and user friendly.
During a comparative analysis of online marketplaces focused on interface design and layout efficiency, I discovered that grid systems are commonly used to enhance clarity and improve browsing flow, which stood out when exploring structured grid shopping portal – The interface presents products in a clean and logical grid arrangement, making it easy for users to explore items without confusion or unnecessary complexity.
During usability testing of ecommerce marketplace systems and browsing flows, I discovered that smooth structure improves user satisfaction, which became clear when exploring simple shopping flow center – The marketplace design feels smooth, and browsing products is fast, easy, and intuitive.
While casually browsing through different platforms, I discovered go to this page – The layout is simple, everything loads quickly, and it feels easy to navigate without delays or confusion.
Many users exploring digital stores appreciate platforms that focus on simplicity and convenience in every stage of shopping where simple buy cart hub is included in descriptions – it highlights an easy to use structure that supports quick browsing, fast selection, and smooth checkout for a more enjoyable online purchasing experience overall.
During a comparative analysis of online marketplaces focused on trust and user experience, I discovered that trusted buying hubs improve satisfaction and browsing efficiency, which stood out when exploring reliable cart shopping hub – The platform looks trustworthy, and navigation is smooth, simple, and easy to follow.
While reviewing serene content websites, I encountered this Elphin Bothy informational hub and the site feels calm, informative, and very pleasant to browse through, with structured content that feels light and easy to absorb.
Felipe prestamos para router alta gama. Teletrabajo sin cortes.
During my search through entertainment pages, I discovered this Chris Young fan page hub and found it pretty engaging overall, keeping me browsing for a while because the content was easy to follow and visually appealing.
Individuals exploring fashion inspiration often turn to online stores that highlight clean design aesthetics and modern clothing collections tailored for expressive personal style Stylish Wardrobe Studio – offering a fashion focused platform that delivers curated clothing selections emphasizing modern aesthetics wearable comfort and design driven apparel suitable for individuals who value both style and practicality in daily outfits
Individuals who enjoy lively shopping experiences often look for platforms that use trading post concepts to create dynamic browsing with varied product selections that make exploration more enjoyable and intuitive Wave Harbor Goods Flow Hub – featuring a structured e commerce platform where trading post design enhances product presentation and creates a dynamic browsing experience that feels organized and visually engaging for users
While reviewing multiple online resources, I stumbled upon explore this ivorycove hub – The layout is pleasant with clear sections, allowing users to explore without confusion or difficulty.
As I continued exploring different online options, I found see this site – The website seemed interesting enough, and browsing through it felt smooth and simple, making navigation easy and comfortable.
While going through community information sites, I noticed this friendly local page and found it very inviting, offering a warm tone that makes the overall browsing experience pleasant and approachable.
In many digital storefront comparisons, users tend to appreciate platforms that maintain order and clarity, especially when they reach Arena CleverCart Explorer – Smooth page transitions and well structured sections contribute to a more engaging browsing flow that keeps users comfortable throughout their visit.
In the process of reviewing digital marketplaces focused on optimized shopping experiences, I found that smart buying frameworks improve usability by creating clear and smooth navigation paths, which became evident when analyzing smart deal finder index – The concept works effectively, making browsing feel simple, structured, and easy for users at all levels.
In the course of evaluating online shopping platforms centered around deals and discounts, I found that clarity in pricing improves user confidence and engagement, which was evident when exploring daily deals showcase portal – The deals section looks attractive, and prices seem reasonable, structured, and easy to compare.
While reviewing several digital vendor platforms for usability and structure, I came across a system that presents its interface in a notably clean way, especially when navigating category pages and product sections centered around Frost Ridge Studio Portal which helps maintain clarity across browsing sessions and reduces visual clutter for users who prefer simple layouts – the interface feels balanced and straightforward, making it easy to locate items without unnecessary complexity.
Many consumers exploring online deals often prefer platforms that offer modern design and structured browsing systems where fast style shopping center is included in descriptions and it highlights a system designed to improve browsing efficiency while ensuring users can easily access products and enjoy a smooth and intuitive shopping journey overall.
Many digital users appreciate websites that feel welcoming and easy to navigate especially when they access Meadow Cotton Goods Outlet – The platform feels soft and organized making browsing simple enjoyable and stress free throughout the entire shopping journey.
While browsing through several online options, I came across take a look here – The structure is nice and balanced, giving a calm and tidy browsing experience that makes navigation comfortable and clear.
During analysis of online retail systems focused on modern UI and polished cart design, I discovered that refined interfaces enhance usability and reduce confusion, which became clear when testing smooth cart experience center – The layout looks sleek and modern, making the shopping experience seamless and user friendly.
Many online shoppers enjoy platforms that frequently refresh their listings with new discounts and rotating offers which makes browsing more engaging when continuous deals loop hub appears within product descriptions and highlights flexible shopping opportunities – it describes a store concept focused on ongoing updates and regularly changing product availability for users who like discovering fresh bargains each time they visit.
While browsing various project-based websites, I came across this Elkhart initiative page and project information seems structured, useful, and clearly presented overall today, with details laid out in a way that feels easy to follow and understand.
As I continued reviewing several websites, I found check this resource – It has a simple and tidy structure where pages feel organized and visually balanced.
People exploring modern apparel often seek fashion platforms that combine aesthetic inspired clothing with stylish and versatile wardrobe collections for daily wear Minimalist Style Clothing Hub – providing a curated selection of modern fashion pieces that reflect clean aesthetics contemporary design and wearable comfort designed for individuals who appreciate simple elegant and trend conscious wardrobe options
During my search through social community sites, I encountered this active team link and liked that it continues to be maintained, showing signs of ongoing updates and steady engagement over time.
Shoppers who value clarity in online stores often choose platforms that organize products using merchant mart layouts where everything is grouped logically into easy to navigate categories Canyon Merchant Goods Hub – offering a well structured shopping environment with category based browsing designed to help users quickly find products while maintaining a clean and efficient online retail experience
During my review of platforms focused on empowerment and awareness, I encountered this important hub and found the content to be well organized, offering a meaningful message that resonates clearly with readers.
During a general browsing session, I encountered check it out – The way everything is arranged makes it easy to explore, with a layout that feels balanced and helps guide users naturally from one section to another.
Shoppers often look for centralized hubs where they can easily compare prices and access multiple discount options without navigating through complex or cluttered websites Value Hunt Marketplace Prime – It provides a simplified browsing experience focused on helping users identify the best deals quickly while maintaining a smooth and organized shopping environment
In today’s competitive online retail landscape, smooth user experience and structured design are crucial when visiting sites such as CartArena Explorer CleverCart Space – A well organized layout helps reduce cognitive load and allows users to focus more easily on browsing products and content.
While reviewing ecommerce systems designed for low pricing and value optimization, I observed that affordable product hubs improve navigation by focusing on savings, which was evident when testing budget savings marketplace hub – The products look reasonably priced and accessible, making it feel like a useful platform for economical shopping.
In analyzing beginner-focused online stores, I found that simplicity improves satisfaction when using systems such as no confusion cart – The shopping cart is designed to eliminate complexity, ensuring users can shop and checkout without any confusion or difficulty.
While exploring various ecommerce platforms focused on gadgets and electronics, I came across a section labeled useful gadget shopping hub – The design presents tech items in an appealing way, helping users discover interesting devices while keeping navigation smooth and product browsing efficient and straightforward.
While navigating through various online resources, I encountered see more here – The browsing experience is smooth overall, with content arranged in a very readable way that supports easy reading.
Many individuals prefer online stores that provide both convenience and variety and while browsing they may see home essentials hub featured in descriptions – it emphasizes accessible products suitable for maintaining household routines and supporting daily living activities effectively.
In various discussions about improving digital storefront usability, contributors often stress the importance of clear navigation, and within that context the embedded reference Granite Store Network offers structured access to marketplace sections, helping users find relevant products more quickly and efficiently.
Many digital shoppers prefer websites that offer warm visual design and intuitive navigation especially when accessing Meadow Cotton Goods Store – The site feels cozy and welcoming making it very easy to browse through different sections without confusion or unnecessary effort during shopping.
In the middle of checking various resources, I discovered check out this page – It uses an interesting design approach, and everything is clear and not overwhelming at all, making browsing simple and comfortable.
People who frequently shop online tend to prefer websites that focus on quick access and wide product selection where fast value shopping center is featured in guides and it highlights a system designed to simplify shopping while ensuring users can quickly discover products and enjoy a smooth checkout process across all categories.
Главные новости: https://gen-web.ru
While browsing conceptual analysis blogs, I noticed truth insight reflection page and content feels reflective, detailed, and somewhat thought provoking overall tone, offering structured ideas that encourage deeper engagement and interpretation. – It feels carefully composed and reflective.
Fashion enthusiasts looking for stylish clothing often prefer brands that offer curated aesthetic collections reflecting modern trends and versatile design approaches Contemporary Wardrobe Fashion Hub – offering a curated fashion platform featuring modern clothing collections and aesthetic inspired designs that emphasize elegance versatility and comfort for individuals seeking refined and expressive personal style choices
Many online shoppers prefer websites that focus on fresh trends and usability especially when accessing Trend Station Digital Market The platform feels like a nice place for trends and products appear updated and quite useful for everyday browsing and discovery of new items.
The updated platform design introduces better structure and improved spacing across all content areas Stone Cotton Simple Portal allowing users to quickly understand layout and access information without difficulty – The experience feels straightforward, minimal, and easy to navigate overall today.
During my browsing session of casual games, I discovered this singles browser game hub and it seems fun and unique compared to typical websites online, providing an unusual but interesting idea that caught my attention briefly.
Thanks for putting in the work to make this approachable, plenty of sites cover the same ground but most do it badly, and a quick visit to epictrendcorner confirmed this one stands apart, simple language and useful examples without anyone trying to sell me anything along the way which I really appreciated.
Shoppers who prefer visually creative online platforms often look for stores that turn product browsing into an artistic experience through curated design and presentation styles Dawn Orchard Boutique Atelier – offering a stylish e commerce platform that emphasizes artistic product display and refined layout design creating a visually engaging and aesthetically pleasing shopping journey for modern users
During some general browsing, I noticed open this link – It offers a nice design style where everything feels structured and easy to navigate intuitively.
As I browsed through high-end travel listings, I noticed this elegant getaway page and appreciated how the visuals and structure create a relaxing, luxurious feeling that makes the destination highly appealing.
Useful information presented in a way that does not feel like a sales pitch, that is what I appreciated most, and a stop at follow this link was the same, no upsell and no fake urgency just steady content laid out properly for someone trying to actually learn from it rather than just be sold to.
During a comparative study of online retail systems focused on open interface design and category accessibility, I discovered that spacious layouts enhance usability and browsing flow, which stood out when analyzing wide browsing shopping portal – The open shopping corner feels roomy and well structured, allowing users to explore categories easily and comfortably.
While comparing several websites for usability, I encountered this useful link – It immediately strikes me as well-designed, presenting a modern appearance that balances visuals and functionality in a way that feels approachable and comfortable to explore.
Shoppers seeking efficient online experiences usually prefer platforms that simplify browsing while ensuring access to reliable sellers where easy seller link center is included in descriptions – it highlights a structured marketplace designed to improve navigation and provide a smooth and trustworthy shopping journey for users across all categories.
While reviewing ecommerce systems designed for cart simplicity and fast checkout, I noticed that direct cart hubs improve clarity and usability, which stood out when exploring smooth purchase cart hub – The system is efficient, offering a fast and clean checkout experience throughout.
Digital shopping experiences are often judged by layout clarity and how quickly users can find desired items Smart Corner Cart Store – The platform offers a pleasant and straightforward browsing experience overall Pages load efficiently and transitions between sections feel smooth and well optimized for usability.
While exploring ecommerce platforms with minimalistic design approaches, I discovered gate access product hub – The browsing experience feels fast and intuitive, with clearly arranged categories that help users navigate efficiently and discover items without being overwhelmed by excessive interface elements.
Users browsing modern e-commerce sites often value clarity and structure that reduce confusion especially on Cove Goods Shopping Zone – The experience felt pleasant and well organized with everything arranged neatly making browsing simple and comfortable throughout the visit.
When reviewing marketplace interfaces, users often focus on responsiveness, and Harbor Vendor Flow – Pages load rapidly and maintain structured layouts, giving users a smooth experience while exploring product categories and comparing different listings across the platform.
During a general browsing session, I encountered check it out – The site is well arranged, and it makes finding items effortless while keeping navigation clear and straightforward.
Individuals exploring fashion inspiration often turn to online stores that highlight clean design aesthetics and modern clothing collections tailored for expressive personal style Stylish Wardrobe Studio – offering a fashion focused platform that delivers curated clothing selections emphasizing modern aesthetics wearable comfort and design driven apparel suitable for individuals who value both style and practicality in daily outfits
Shoppers searching for reliable e-commerce platforms often value websites that provide fast navigation and clear product organization where smart shopping easy hub appears in content and it reflects a system designed to enhance usability while helping users quickly browse products and complete purchases without unnecessary complications or confusion in the process.
While browsing local event websites, I came across this Edinburgh event guide page and event information looks lively, well organized, and easy to understand, with details presented in a clear and accessible way that makes planning feel simple.
Many shoppers appreciate platforms that provide fast cart interaction and smooth navigation especially when they land on Cart Dynamic Zone Hub The shopping experience feels smooth and the cart system works really fast today ensuring quick and easy browsing across all product categories.
In the middle of checking different platforms, I came upon discover this honeycove studio site – Fast loading pages and clean layout make browsing quite comfortable with a smooth structure.
During my review of regional discovery sites, I encountered this Ireland travel insights page and thought it was helpful, especially for someone new to this topic, as it provides easy-to-follow and informative guidance about the area.
A recent usability review highlights that the updated design improves spacing and visual hierarchy throughout the interface Cottonstone Explorer Site making it easier for users to scan content and identify key sections without hesitation or confusion – Overall structure is cleaner and more user friendly, supporting better engagement across all pages today.
While analyzing ecommerce UX designs centered on clean layouts and user comfort, I observed that fresh hub systems improve usability and reduce visual strain, which was evident when reviewing refreshing shopping experience hub – The design feels light and well arranged, making browsing smooth and enjoyable.
As I explored various creative pages earlier, I noticed a unique site reference which impressed me because the visual hierarchy is well balanced and the overall aesthetic gives off a professional yet welcoming vibe that feels carefully crafted.
Shoppers who enjoy structured online marketplaces often prefer platforms that use a goods district concept where products are grouped in a clean and organized layout making browsing feel simple intuitive and efficient for everyday purchasing needs Golden Cove District Market Hub – offering a goods district inspired shopping experience designed with clear organization and easy navigation so users can explore items effortlessly while enjoying a smooth and logically structured online browsing environment
Many online shoppers appreciate platforms that simplify navigation and reduce friction when purchasing everyday products, and one example is FlowBuy Hub – The website is designed to make purchasing feel seamless and intuitive, helping users move through categories quickly while keeping the overall experience smooth and efficient for regular browsing sessions.
Online users often search for platforms that enhance trust by connecting them with verified sellers and dependable product sources where trusted vendor link portal is mentioned in content focused on reliability – it highlights a system designed to improve user confidence while ensuring seamless navigation and efficient purchasing across a wide range of products.
While evaluating ecommerce platforms focused on structured shopping environments and user-friendly navigation, I noticed that organized marketplaces significantly improve product discovery and browsing efficiency, which became clear when exploring organized buying zone hub – The marketplace is well structured, and products are easy to locate, making the overall shopping experience smooth and efficient.
While exploring modern ecommerce platforms focused on centralized shopping systems and category variety, I noticed that ultra hub designs often improve user experience by bringing many product types together in one place, which became clear when analyzing modern ultra shopping hub – The ultra hub feels very modern and well organized, offering a wide range of categories in one place that makes browsing simple, efficient, and easy for users across different shopping needs.
While casually browsing through different platforms, I discovered go to this page – The site feels useful, and the content is shown in a clear way that makes navigation smooth and easy to follow.
Shoppers frequently prefer platforms that offer clarity and structured content especially when accessing Goods Ridge Silk Atelier Hub Clean design and smooth navigation made this a pleasant visit helping users stay focused and move easily through different product categories.
Came across this through a roundabout path and now it is on my regular rotation, and a stop at view this website sealed that decision, the open web still produces serendipitous discoveries when you let the citations and references guide you rather than relying purely on algorithmic feeds for new content recommendations always.
Users engaging with modern e-commerce websites often value simplicity and performance, especially when accessing CleverCart Zone Portal – The layout is well organized, loading speed is reliable, and navigation supports a smooth and efficient browsing experience across all devices.
Across evaluations of online shopping ecosystems and interface design practices, consistency is frequently highlighted, and Cove Market Craft Exchange appears in discussions about streamlined browsing – the overall experience is smooth, with well-arranged categories that allow users to locate products quickly and comfortably.
Individuals searching for fashion inspiration often turn to platforms that emphasize aesthetic clothing collections and modern design focused apparel for everyday wear Contemporary Aesthetic Wear Hub – offering a curated selection of stylish clothing that blends modern fashion trends with elegant simplicity and versatile wardrobe pieces designed for expressive personal styling and daily comfort
A memorable post for me on a topic I had thought I was tired of, and a look at take a look here suggested the same site can refresh other tired topics, sites that can revive my interest in subjects I had written off as exhausted are doing rare work and this one is clearly doing that for me today.
While reviewing different online platforms, I stumbled upon explore this site – The site is well organized and maintains a consistent and visually appealing presentation throughout all pages.
During some light online research, I ran into view this link – The interface feels smooth and well built, and it is easy to explore content while maintaining a clear and simple layout.
While exploring advocacy and informational websites, I came across this Florida Can Do Better page and the website presents optimistic messaging, clear goals, and simple layout design, making the content feel approachable and easy to understand at a glance.
Many consumers browsing online stores often appreciate platforms that offer multiple deal options and easy product discovery where budget deal access hub is included in guides and it highlights a system designed to streamline shopping while ensuring users can quickly locate affordable products and enjoy a smooth and intuitive experience overall.
Many online shoppers prefer platforms that highlight fair pricing and strong offers especially when accessing DynamicDeal Center Market The deals here are attractive and prices feel fair and competitive online helping users shop confidently without worrying about overpricing or unclear listings.
Нужен займ? займ для студентов мгновенное решение, перевод средств и минимум требований. Идеально для срочных финансовых ситуаций и быстрых расходов
During my search through creative web pages, I encountered this modern concept hub and I like the overall presentation, feels modern and straightforward, with a design that feels clean, minimal, and easy to navigate.
In the course of evaluating online retail platforms focused on clarity and usability, I found that core store designs improve browsing efficiency by simplifying layouts, which was evident when analyzing clean core shopping portal – The design appears minimal and organized, with product listings that are easy to read and understand.
The platform’s refreshed layout provides clearer content grouping and improved accessibility across different browsing sections Creekstone Heritage Market with faster response times and better usability overall today – Users find it easier to engage with content, thanks to a more organized and intuitive interface structure.
In the process of analyzing different outreach websites, I discovered this clear presentation which delivers its content in a logical sequence, ensuring that visitors can easily follow the narrative from beginning to end without confusion.
Online users who enjoy efficient shopping experiences frequently look for platforms that eliminate delays and simplify transaction processes where quick commerce gateway hub appears in guides focused on usability – it highlights a streamlined system that helps users move quickly from product discovery to final purchase with minimal effort required.
While reviewing multiple online resources, I stumbled upon explore this brook foundry site – The interface is easy to use, and all elements are placed in logical sections that make navigation clear and direct.
Нужен эвакуатор? эвакуаторы в солнечногорске вызвать быстрая помощь на дороге 24/7. Перевозка автомобилей любой сложности, доступные цены и оперативный выезд по городу и области
People who enjoy structured online shopping often look for platforms that combine a commerce hub layout with smooth navigation and a wide selection of products making it easier to explore different categories in one place Ridge Velvet Market Commerce Hub – providing a clean e commerce experience where the hub design supports easy browsing and wide product availability ensuring users can navigate smoothly while discovering items across all categories in a simple layout
Сломалась машина? вызвать эвакуатор принимаем заказы онлайн круглосуточная работа, быстрый приезд и аккуратная транспортировка авто. Помощь при ДТП, поломках и срочных ситуациях
While analyzing ecommerce UX designs focused on daily necessities and usability, I observed that practical organization enhances user satisfaction, which was evident when reviewing essential daily hub – The platform is simple and efficient, offering well categorized everyday products for easy shopping.
During analysis of digital shopping experiences emphasizing user control, I observed a section labeled adaptive choice shopping hub – The design allows flexible browsing and selection, helping users explore different product options and choose items smoothly while maintaining a clean and organized interface overall.
Online users often prefer e-commerce websites that maintain clarity and intuitive flow especially when they access Guild Raven Grove Retail Hub The layout feels interesting and user friendly making browsing simple and efficient while helping users easily find what they are looking for.
Fashion oriented users often seek brands that highlight stylish and minimalist clothing collections designed with modern aesthetic influences for everyday outfits Stylish Aesthetic Apparel Hub – offering a curated fashion platform featuring elegant clothing collections and contemporary designs that focus on comfort style and versatility for individuals who appreciate modern wardrobe aesthetics
While exploring different online platforms earlier today, I came across check this page – The pages load fast overall, and the experience feels responsive and user friendly, making browsing smooth and efficient without any noticeable delays.
When reviewing e-commerce platforms focused on vendor aggregation, analysts often note that clarity in categorization significantly improves the overall shopping experience for both new and returning users alike Stone Harbor Vendor Showcase – the interface is considered efficient and well designed, allowing visitors to browse comfortably while quickly identifying relevant product options across different sections.
While checking out several online resources, I stumbled upon click here now – I gave it a visit and everything navigated properly, with no issues or delays while accessing different parts of the site.
When browsing online shopping hubs, users often value simplicity and fast response times, particularly while visiting DealSpot Clever Station – The website maintains a clean interface that supports easy navigation and helps users focus on finding relevant deals without unnecessary complexity or confusion.
While exploring light comedy reference archives and simple humor sites, I came across light comedy reference archive and the page feels minimal, easy to browse, and gently humorous, making it suitable for quick and casual viewing. – A simple and mildly entertaining web concept.
Found a small mental shift after reading this, the framing here is just a bit different from the standard takes online, and a look at futurecartcorner extended that fresh perspective across more material, the rare site whose voice actually changes how you think about something rather than just confirming existing beliefs.
Many consumers browsing online stores often appreciate platforms that highlight diverse products and simple navigation tools where easy world shopping hub is included in guides and it highlights a system designed to improve usability while ensuring users can quickly find products and complete transactions without unnecessary effort or confusion.
Online customers browsing through curated deals and new arrivals across different categories often come across storefront navigation link interwoven references that help them move smoothly between sections enhancing the overall experience of discovering products without losing browsing context mid session – The flow between categories feels natural and well structured.
In the course of evaluating online retail platforms focused on navigation flow and structure, I found that line-based shop systems enhance browsing clarity and speed, which was evident when analyzing linear browsing store hub – The design looks well organized, making it easy to locate items across categories.
In the process of browsing regional guides online, I came across this coastal data information page and it seems like a niche site but still quite informative overall, offering structured content that is surprisingly clear for such a niche topic.
During a casual browsing session across various websites, I noticed open this commerce atelier site – It features a clean and modern design that makes exploring content feel effortless overall and very easy to follow.
Quietly impressive in a way that does not announce itself, and a stop at check out this page extended that quiet impressiveness, the kind of quality that emerges through sustained attention rather than first impressions is the kind I trust more deeply and this site has been earning that deeper trust across multiple sessions over time consistently.
Modern online shops benefit greatly from cohesive design systems and fast responsiveness, as demonstrated when users access < Drift Orchard Marketplace – The browsing experience remains fluid, content loads efficiently and the overall layout supports easy exploration.
Online buyers who prefer structured shopping platforms often choose stores that simplify navigation and improve deal discovery where t gadget value marketplace zone allows users to browse technology accessories easily while focusing on affordability and practical product selection for daily use and device enhancement purposes.
During my review of fine jewelry platforms, I encountered this luxury collection site and found it visually impressive, as the layout feels polished and carefully arranged to highlight products in a very classy and professional manner.
Many digital buyers appreciate platforms that balance product variety with trustworthy service, especially when shopping regularly, and this is reflected in Trusted Dock Bazaar – The marketplace is designed to support smooth navigation and reliable purchasing options so users can explore goods easily while enjoying a consistent online shopping experience.
During analysis of online retail platforms focused on direct buying functionality, I discovered that streamlined flows enhance user experience and reduce friction, which became clear when testing direct shopping experience hub – The platform feels efficient, with clear navigation and a simple purchasing process.
During an analysis of ecommerce UX improvements focused on speed, I observed a section labeled quick view retail gateway – The platform is highly responsive, allowing pages to load instantly and making browsing feel fluid and efficient for users who prioritize fast and seamless shopping experiences.
Нужна эвакуация машины? эвакуатор зеленоград дешево круглосуточно телефон эвакуатор быстрое реагирование, аккуратная погрузка и безопасная доставка автомобиля в нужное место
Individuals exploring fashion inspiration often turn to online stores that highlight clean design aesthetics and modern clothing collections tailored for expressive personal style Stylish Wardrobe Studio – offering a fashion focused platform that delivers curated clothing selections emphasizing modern aesthetics wearable comfort and design driven apparel suitable for individuals who value both style and practicality in daily outfits
In modern online shopping environments users frequently prioritize clarity and performance especially when visiting Orchard Drift Market Place Pages load fast and the content is displayed in a structured way making browsing simple efficient and user friendly while ensuring a smooth experience across all sections of the platform.
People who enjoy clear and structured online shopping environments often appreciate platforms that focus on elegant design and organized product presentation for easier navigation Ridge Elegant Product Hub – featuring a clean and refined online store where products are showcased with structured layout design and visual simplicity creating a smooth and enjoyable browsing experience for all users
In the middle of checking various resources, I discovered check out this page – The design feels clean and organized, and nothing appears cluttered while moving through sections, which helps keep browsing smooth and simple.
During my search through cinema showcase platforms, I discovered this Folie a Deux film artwork site and film related content appears artistic, engaging, and thoughtfully presented style, with visuals that feel creative, immersive, and thoughtfully designed.
When reviewing platforms that combine learning with creativity tools, many highlight the importance of accessibility and interactive design elements that support engagement Outlet Creative Navigator – users appreciate how the site encourages experimentation and idea building through an intuitive layout that makes content discovery feel both simple and enjoyable.
While navigating through various online resources, I encountered see more here – The layout is simple and clear, and pages are straightforward and easy to read, ensuring a stress free experience.
Many digital shopping environments succeed when they reduce friction and improve usability, especially when users visit Goods Arena Efficient Store as it helps them browse smoothly and find relevant items without unnecessary delays or complications.
While conducting comparative research on ecommerce UX and checkout systems, I observed that flexible cart options improve browsing continuity and purchase flow, which was clear when testing smart purchase cart portal – The cart feels versatile, and the checkout process is simple, fast, and user friendly.
During a quick search session, I noticed this useful link – The site looks straightforward, and while moving through pages, everything stayed clear and easy without causing confusion.
Many users exploring digital shopping platforms often enjoy simplified browsing when they discover tools like dynamic goods browsing hub which organizes products clearly and helps users navigate categories with ease while comparing options efficiently online for better user experience today smoothly
Портал для туристов https://aliana.com.ua для путешественников: направления, маршруты, советы и лайфхаки. Подбор отелей, билетов и экскурсий, идеи для отдыха и полезные рекомендации. Планируйте поездки легко и открывайте новые страны с комфортом.
Consumers frequently searching for online deals tend to prefer platforms that make shopping faster and more intuitive where smart nest shopping center appears in guides and it reflects a structured system designed to simplify browsing while helping users compare products efficiently and complete transactions without unnecessary confusion or delays in the process.
While browsing online information directories, I came across this OneCentral resource portal and got a good first impression, as the content appears organized and useful, making it easy to understand what the site offers at a glance.
Anyone curious about this topic would do well to start here, the foundation laid is solid, and a stop at futuregoodszone would round out their understanding nicely, this is the kind of resource I would point a friend toward without hesitation if they asked me where to begin learning about anything in this area.
Online buyers searching for efficient deals usually prefer marketplaces that offer fast browsing tools and well structured product categories for easier shopping rapid checkout marketplace hub supporting quick transactions and reducing time spent during the purchasing process – It demonstrates the value of speed focused design in modern e-commerce platforms.
As I explored online business improvement content, I came upon this useful business hub and liked how it provides practical suggestions that feel meaningful and directly applicable to real-world scenarios.
The platform now emphasizes smoother content flow, allowing users to transition between pages with greater ease and reduced confusion Caramel Cove Visual Archive – The browsing environment feels more structured, with improved layout flow that enhances readability and user engagement across pages.
While conducting usability evaluations of ecommerce platforms focused on structured design and clarity, I noticed that clean shopping hubs improve user experience and navigation flow, which stood out when reviewing simple shopping structure portal – The browsing system is clean, and moving between sections feels smooth and intuitive.
While reviewing ecommerce platforms optimized for browsing flow and usability, I noticed that stacked interfaces make product discovery more intuitive by reducing clutter and improving structure, which stood out when exploring smart layered stack index – The design feels smooth and easy to navigate, allowing users to scroll through products effortlessly and efficiently.
People who enjoy aesthetic fashion often look for curated clothing brands that combine modern design elements with stylish and versatile apparel collections Modern Style Apparel Hub – offering a fashion platform that features elegant clothing collections inspired by contemporary aesthetics and designed for individuals seeking comfortable stylish and expressive wardrobe options for everyday use
During some general browsing, I noticed open this trading link – It offers a smooth experience overall, and navigation feels quick and very user friendly, making the interface easy to use.
In digital shopping environments users frequently prioritize clarity and usability especially when visiting Frost Goods Seaside District The layout looks clean and structured making browsing simple and efficient while ensuring users can easily navigate through different product sections without difficulty.
Worth recommending broadly to anyone who reads on the topic, and a look at go to this page only confirms that, the rare combination of accessibility and depth in this site makes it suitable for both newcomers and people who already know the area which is hard to pull off in any blog format today and rarely managed.
As I continued exploring different websites, I found see this site – It is well structured, and the navigation feels natural and very straightforward, creating a smooth and easy browsing experience overall.
During analysis of online retail platforms focused on speed and responsiveness, I discovered that quick-loading interfaces enhance user experience and reduce friction, which became clear when testing fast flow shopping portal – The platform responds quickly and offers a smooth, well optimized shopping corner.
People looking for efficient online shopping often choose platforms that focus on simplicity and organized layouts to make browsing and purchasing more convenient and stress free Bay Harbor Merchant Space – providing a straightforward digital marketplace experience centered on practical browsing easy navigation and quick access to essential products designed to improve everyday shopping efficiency
Актуальний сучасний український журнал Різні це джерело натхнення, новин і корисних матеріалів. Читайте статті про життя, тренди та розвиток у зручному форматі
While exploring unique fashion resale edits, I discovered unique preloved fashion edit and the collection feels expressive, offering distinctive pieces that stand out while remaining tastefully arranged. – It feels creative, refined, and visually engaging today now experience here view today check browse.
Online shoppers often value platforms that reduce friction, load quickly, and maintain clear visual hierarchy when presenting multiple product categories across a single interface CGS Shopping Hub – A smooth shopping environment ensures users can find what they need quickly while enjoying a visually balanced and intuitive browsing experience throughout the site.
When people explore online learning tools designed for innovation and brainstorming, they often prefer platforms that combine structure with creative flexibility and ease of use Value Creative Flow Portal – the site delivers a smooth experience that encourages curiosity and supports users in building and refining their ideas through intuitive interaction design.
People browsing for efficient shopping experiences often encounter Product picking dashboard which centralizes item selection and helps users view multiple options at once making comparisons easier while maintaining a clear and structured interface designed for faster decision making – improves overall browsing efficiency significantly.
While navigating through various online resources, I encountered see more here – The platform feels clean and easy to use, with a simple structure that allows quick understanding of the layout and content.
Many consumers exploring online deals often prefer platforms that offer simple navigation and fresh discount updates where fast discount corner center is included in descriptions and it highlights a system designed to improve browsing efficiency while ensuring users can easily access products and enjoy a smooth and intuitive shopping journey overall.
Shoppers who prioritize trust in online purchasing usually prefer platforms that guarantee safety and responsive service where trusted shopping assistance center is featured in guides and it reflects a marketplace designed to ensure secure transactions while providing reliable support and maintaining user satisfaction throughout the entire shopping experience.
During my search for uplifting content online, I encountered this positivity message hub and really like the message behind this, feels genuine and supportive, presenting ideas that feel honest, caring, and emotionally reassuring overall.
mooncovegalleria.shop – Nice structure and clarity, everything is easy to find quickly.
While conducting comparative analysis of ecommerce cart solutions and checkout structures, I observed that functional design improves user experience and reduces friction, which was evident when reviewing efficient shopping cart center – The cart solution looks functional, and the shopping steps are simple, clear, and easy to follow.
Users of online shopping platforms often look for sites that combine ease of use with organized product categories, and such a site is which offers a structured browsing experience designed to support quick decisions and simple navigation for shoppers Premium Deck Store which offers a structured browsing experience designed to support quick decisions and simple navigation for shoppers – It focuses on providing a smooth and practical online shopping environment
Recent design updates provide better spacing and more logical grouping of elements, where Crystal Cove Visual Collection Portal is embedded mid content, while the browsing environment feels stable, clean, and very easy to use.
Individuals exploring fashion inspiration often turn to online stores that highlight clean design aesthetics and modern clothing collections tailored for expressive personal style Stylish Wardrobe Studio – offering a fashion focused platform that delivers curated clothing selections emphasizing modern aesthetics wearable comfort and design driven apparel suitable for individuals who value both style and practicality in daily outfits
Felt the writer was speaking my language without trying to imitate it, and a look at view this website continued that natural fit, when a writers default voice happens to match what you find easy to read the experience feels frictionless and that is something I notice and remember about specific sites going forward.
During a comparative study of digital retail platforms focused on navigation clarity, I found a page labeled structured shopping port center – The interface uses a clean port inspired design that makes browsing intuitive, ensuring users can easily move between categories and understand product organization at a glance.
Online users often value platforms that provide clarity and structured presentation especially when they land on Harbor Marketplace Flow Birch The design is well organized and I like how it makes exploring very convenient while keeping navigation easy and intuitive throughout.
Нужны срочно деньги? займ 30000 на карту подайте заявку онлайн и получите деньги в кратчайшие сроки с прозрачными условиями и удобным погашением
While analyzing ecommerce platforms designed for performance-driven cart systems and user convenience, I observed that efficient cart solutions improve satisfaction and reduce friction, which became evident when testing quick checkout solutions hub – The cart experience feels smooth and efficient, allowing users to browse and complete checkout without unnecessary delays.
Если вам нужна профессиональная верификация GMB в условиях российских ограничений — обратитесь к специалисту напрямую.
During my review of scenic accommodation websites, I noticed this Fisherman’s Retreat getaway listing site and the location feels peaceful, scenic, and ideal for relaxing stays experience, presenting a serene environment that feels inviting and restful.
Shoppers who enjoy curated online spaces often appreciate platforms designed like boutique halls where products are carefully selected and displayed in a simple structured layout that makes browsing feel more refined and easy to navigate Glade Ridge Boutique Hall Market – offering a boutique hall inspired shopping experience with clean organization and curated product presentation designed to help users explore items effortlessly while enjoying a smooth and visually pleasant online shopping environment
In competitive e-commerce environments, usability and visual balance play a major role in user satisfaction, especially when accessing CleverZone Product Space where categories are arranged clearly, allowing visitors to move smoothly through listings without feeling overwhelmed or distracted by unnecessary design complexity.
Worth recognising the specific care that went into how this post ended, and a look at fastcartcenter maintained the same careful conclusions, endings are where most blog content falls apart and this site has clearly invested in the closing stretches of its pieces rather than letting them simply trail off when energy fades.
When examining online retail platforms designed for improved user experience, analysts often highlight smooth transitions between categories and clearly structured product displays when interacting with Vendor Atelier Hazel Harbor Space – Smooth browsing experience, products are clearly displayed and accessible, offering users a straightforward path to explore items while maintaining an intuitive and visually balanced layout throughout the site.
While exploring several platforms online, I stumbled upon explore here – The layout feels nicely structured, with everything looking clean and thoughtfully arranged in a way that enhances the overall browsing experience.
As I browsed through informational campaign sites, I encountered this Simpsons-related Stride page and this looks meaningful and worth spending some time exploring, with a layout that feels structured and content that seems purposeful and readable.
Reputable service can you buy a monetized youtube channel publishes detailed product cards showing account age, verification status, included assets, and exact pricing tiers. Aged profiles with natural activity patterns consistently outperform fresh registrations in ad delivery quality and checkpoint avoidance rates. Whether you need accounts for testing or production campaigns, the catalog covers every tier from entry-level to premium.
roulett online, [url=http://hempelpromome.com/index.php/2026/04/08/roulette-mit-echtgeld-strategien-und-tipps-fur/]http://hempelpromome.com/index.php/2026/04/08/roulette-mit-echtgeld-strategien-und-tipps-fur/[/url] stellt dar, eine fesselnde Art des GlГјcksspiels. Spiele beinhaltet in zahlreichen Varianten und locken zahlreiche Spieler. Tauche ein in den Nervenkitzel des Spin.
Top-rated dealer tik tok advertiser account has been serving the media buying community since 2020 with consistent product quality and responsive customer support. Transparent replacement policy covers the first-login window and ensures buyers receive exactly what is described on the product card. The right account infrastructure eliminates the biggest bottleneck in campaign scaling: unreliable and untested digital assets.
While analyzing ecommerce UX systems designed for improved browsing efficiency, I noticed that smart hub models simplify navigation and improve product discovery, which stood out when testing intelligent commerce hub index – The smart hub approach makes shopping efficient and structured, helping users browse smoothly and find products without unnecessary effort.
Users exploring the updated layout report improved clarity across menus and product sections while navigating smoothly Cotton Meadow Trading Base – Navigation appears more intuitive overall, with reduced clutter and better spacing that helps visitors move through pages effortlessly and understand content hierarchy at a glance.
During a comparative study of online retail systems emphasizing simplicity and usability, I discovered that clean cart layouts enhance user experience and navigation clarity, which stood out when analyzing easy cart shopping portal – The platform uses a simple structure that keeps shopping straightforward and removes any confusion while moving through pages.
During my review of online stores, I encountered this product discovery page and found some appealing products, with navigation feeling fast and convenient, making the overall shopping process smooth and easy to handle.
Many shoppers appreciate platforms that keep interfaces light and functional especially when they access Marble Cove Commerce Flow Hub The design is simple but effective and it works really well for quick browsing providing a smooth and stress free user experience overall.
Worth bookmarking and sharing with anyone interested in the topic, that is my honest take, and a stop at futuretrendzone reinforces that, the kind of generous resource that makes the open web feel worth defending against the constant pressure to retreat into walled gardens and curated feeds today everywhere I look across all my devices.
People who enjoy organized e commerce platforms often look for websites that use merchant lane systems to divide products into clear sections making browsing more intuitive and efficient for everyday shopping needs Coast Maple Goods Lane Market – providing a structured online shopping experience with a merchant lane layout that enhances navigation and helps users explore product categories easily while maintaining a clean and user friendly browsing environment
In digital retail environments users often prioritize clear structure and trendy design elements that support easy navigation, particularly on Trend Flow Center – The browsing system is smooth and visually appealing, allowing users to explore products effortlessly while maintaining a clean and modern interface throughout their visit.
When studying digital storefront usability across emerging marketplace technologies and evolving consumer behavior patterns in online environments HoneyCove Shopping Experience Lab researchers consistently find that structured layouts and predictable navigation significantly improve task completion rates – users describe the platform as intuitive and easy to navigate during extended browsing sessions.
While analyzing ecommerce platforms optimized for smart structure and category clarity, I noticed that organized marketplaces improve browsing flow and product accessibility, which stood out when reviewing smart goods discovery hub – The products are well categorized, making the shopping experience smooth and intuitive.
Online shopping experiences improve significantly when platforms prioritize usability and clear structure, allowing users to find products faster and with less effort, and this is the goal of ValuePath Market – It ensures a smooth flow from browsing to checkout while maintaining a focus on affordability and practical product selection for everyday needs
While analyzing ecommerce design patterns for user experience quality, I discovered comfort browsing product hub – The layout provides a cozy and well organized structure that makes product discovery easy and enjoyable for users who prefer a relaxed and simple shopping environment overall.
Reliable source aged facebook account buy connects advertisers with thoroughly vetted profiles backed by replacement guarantees and dedicated support. Product cards display exact specifications including account age, verification level, included assets, geo origin, and current stock availability. Marketplace standards ensure that every account performs as described — no surprises at checkout, login, or campaign launch.
Honestly enjoyed not being sold anything for the entire duration of the post, and a look at fastgoodsarena kept that pleasant absence going across more pages, content that exists for its own sake rather than as a funnel to a paid product is increasingly rare and worth supporting where I can find it.
Reviewing the latest site adjustments, many users appreciate the smoother transitions between pages, and within the explanatory section there is Serene Coastal Outpost included naturally, while surrounding notes mention enhanced layout spacing, clearer visual hierarchy, and a more relaxed browsing experience that encourages longer and more comfortable sessions on the platform.
Dedicated platform where can i buy reddit account helps performance teams find the right account infrastructure for scaling their advertising operations efficiently. The platform combines speed and reliability Ч most products are delivered automatically within minutes after payment confirmation. Professional media buying starts with professional tools Ч source from a marketplace built by advertisers, for advertisers.
Many users browsing digital stores value clarity and intuitive navigation especially when they visit Vendor Harbor Collective Stone Hub The browsing experience felt smooth and nothing was confusing while navigating which made everything easy to find and explore comfortably.
A piece that built up gradually rather than front loading its main points, and a look at check out this page maintained the same gradual structure, content that trusts the reader to reach conclusions through accumulating reasoning is more persuasive than content that announces conclusions and then defends them and this site uses the persuasive approach.
While exploring different sites for information, I landed on take a look and checked it out today, noticing that everything was well structured with easy navigation and clear content flow.
Individuals who enjoy visually active online stores often look for platforms that use trading post layouts to create dynamic browsing experiences with varied product selections that improve engagement and shopping enjoyment Wave Harbor Product Trading Hub – featuring a structured e commerce platform where trading post design enhances product variety and creates a dynamic browsing flow that supports smooth navigation across multiple shopping categories
Online shoppers often prefer platforms that provide clarity and intuitive structure for better navigation, particularly when accessing Trend Corner Flow Market – The website design supports easy browsing and helps users discover trending products quickly while keeping the experience smooth and visually balanced overall.
During usability testing of online shopping systems focused on discount presentation and value clarity, I discovered that deal platforms improve engagement by emphasizing savings opportunities, which became evident when exploring smart price savings hub – The deals appear attractive and well highlighted, giving the impression of a platform that is great for budget friendly shopping.
While exploring niche e-commerce communities and independent seller platforms, I encountered a reference to Elm Harbor curated shop hub which seems to compile vendor listings and product showcases in one accessible place enabling users to browse and compare offerings efficiently – I found it reasonably helpful for discovering curated products
HoneyCoveVendorStudio – Clean interface, everything loads fast and works very smoothly.
I reviewed a number of outlet browsing platforms and encountered a page that appeared well structured and simple Hollow Creek directory access making it easy to move through different sections – It feels practical and straightforward for casual users with smooth experience overall use
While browsing community motorsport events, I came across charity racing support page and interesting concept overall, seems well organized and quite engaging today, presenting information in a way that feels clear, purposeful, and easy to understand. – The structure feels thoughtful and engaging.
Testing results show that the interface now supports better content flow and improved readability, especially around areas where Chestnut Harbor Trade Network is integrated naturally into the text, while usability feedback remains positive – The structure is straightforward, helping users understand page hierarchy easily and browse without confusion or unnecessary effort across different sections of the site.
During research on platforms that improve online shopping efficiency, I found simple shopping index offering structured product listings – this shopnetmarket.shop interface emphasizes clarity, helping users move through categories quickly while maintaining a clean layout that makes browsing deals and selecting relevant items easier for everyday online purchasing decisions.
Полная статья здесь: https://ekostroy76.ru
Магазин бытовой химии https://bytovaya-sfera.ru большой выбор средств для уборки, стирки и ухода за домом. Качественная продукция, доступные цены и быстрая доставка
Срочный онлайн займ https://buhgalter-uslugi-moskva.ru быстрое решение финансовых вопросов. Оформление за несколько минут, высокий шанс одобрения и перевод денег на карту без лишних документов
A particular kind of restraint shows up in the writing, and a look at check out this website maintained the same restraint across pages, knowing what not to say is just as important as knowing what to say and this site has clearly developed strong instincts on both sides of that editorial line throughout pieces I have read.
In the course of evaluating online retail platforms focused on worldwide cart integration and product diversity, I found that global cart systems enhance usability and selection efficiency, which was evident when analyzing global browse cart center – The platform uses a global cart approach, offering a wide selection of online products in one convenient space.
During a light browsing session, I checked out click to view and noticed the site had clean content layout and easy navigation that made the overall experience quite smooth and pleasant.
Shoppers who enjoy peaceful online shopping environments often prefer platforms that use lakefront lounge aesthetics to create a smooth browsing flow and visually pleasant product display experience Velvet Lakefront Elegant Market Hub – offering a structured e commerce platform where calming lakefront design enhances product presentation and ensures a relaxed browsing experience that feels visually balanced and easy to navigate
While exploring niche e-commerce communities and independent seller platforms, I encountered a reference to Elm Harbor curated shop hub which seems to compile vendor listings and product showcases in one accessible place enabling users to browse and compare offerings efficiently – I found it reasonably helpful for discovering curated products
While reviewing e-commerce platforms focused on affordability and usability, I found hyper shopping corner hub which presents products in an organized layout – Hyper Cart Corner offers a really easy shopping experience with affordable products and fast delivery, helping users enjoy smoother navigation and faster access to relevant deals across multiple product categories.
Shoppers often prefer websites that combine modern design with simple navigation systems that enhance usability, particularly on TrendMart Easy Store – Everything is well structured, allowing users to enjoy a smooth browsing experience while quickly locating products across multiple categories with ease.
Reading this brought back the satisfaction I used to get from blogs ten years ago, and a stop at globalcartcorner kept that nostalgic quality alive, sites that capture what was good about an earlier era of internet writing are increasingly precious and this one is doing that without feeling like a deliberate throwback at all.
Digital shopping environments are becoming more competitive as platforms strive to offer better deals and faster delivery options worldwide SmartEdge Bargain Center – This marketplace prioritizes affordability and speed, ensuring users can access promotions quickly and complete purchases efficiently.
When assessing online marketplace usability improvements aimed at simplifying browsing behavior and enhancing product visibility across structured digital catalogs Foundry Icicle Vendor Hub users describe smooth interaction – Overall experience feels clean and efficient with intuitive navigation minimal clutter and well organized sections that make browsing and comparing products easy and comfortable.